WWelcome to the Research Laboratory of Advanced Materials Chemistry at the University of Crete. Our research focuses on the design, synthesis, structural characterization, and application of advanced nanostructured and nanoporous materials for electrocatalysis, photocatalysis, energy conversion, and environmental technologies.
A central direction of our current work is the development of functional materials for alkaline water electrolysis, the oxygen evolution reaction (OER), and green hydrogen production. We design metal oxide, metal chalcogenide, and nitride-based frameworks with controlled pore architecture, surface chemistry, interfacial charge transfer, and physicochemical properties.
Our approach combines wet-chemical synthesis, solid-state methods, electrochemical analysis, and operando spectroscopic techniques to elucidate catalytic mechanisms and enhance performance in sustainable electrochemical energy systems.
Posted by Owner
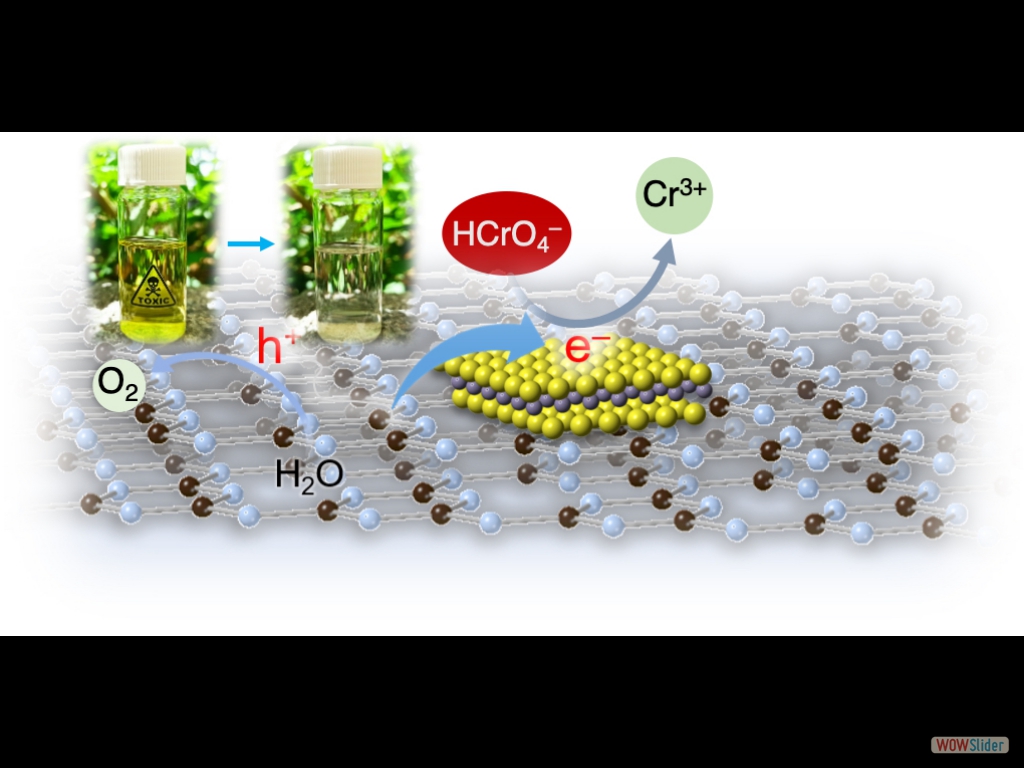 In this article, we report the synthesis of 2D/2D SnS₂/g-C₃N₄ layered heterostructures with reduced interfacial resistance and improved charge transfer kinetics via a photochemical deposition method. The SnS₂/g-C₃N₄ catalysts demonstrate outstanding photocatalytic Cr(VI) reduction (with a 21.2 μmol h⁻¹ Cr(VI) consumption rate) and water oxidation (with a 15.1 μmol h⁻¹ O₂ evolution rate) activity, reaching energy conversion efficiencies of up to 16.4% and 12.1% at 375 nm and 410 nm, respectively, that is among the best known Cr(VI) reduction catalysts reported to date.
In this article, we report the synthesis of 2D/2D SnS₂/g-C₃N₄ layered heterostructures with reduced interfacial resistance and improved charge transfer kinetics via a photochemical deposition method. The SnS₂/g-C₃N₄ catalysts demonstrate outstanding photocatalytic Cr(VI) reduction (with a 21.2 μmol h⁻¹ Cr(VI) consumption rate) and water oxidation (with a 15.1 μmol h⁻¹ O₂ evolution rate) activity, reaching energy conversion efficiencies of up to 16.4% and 12.1% at 375 nm and 410 nm, respectively, that is among the best known Cr(VI) reduction catalysts reported to date.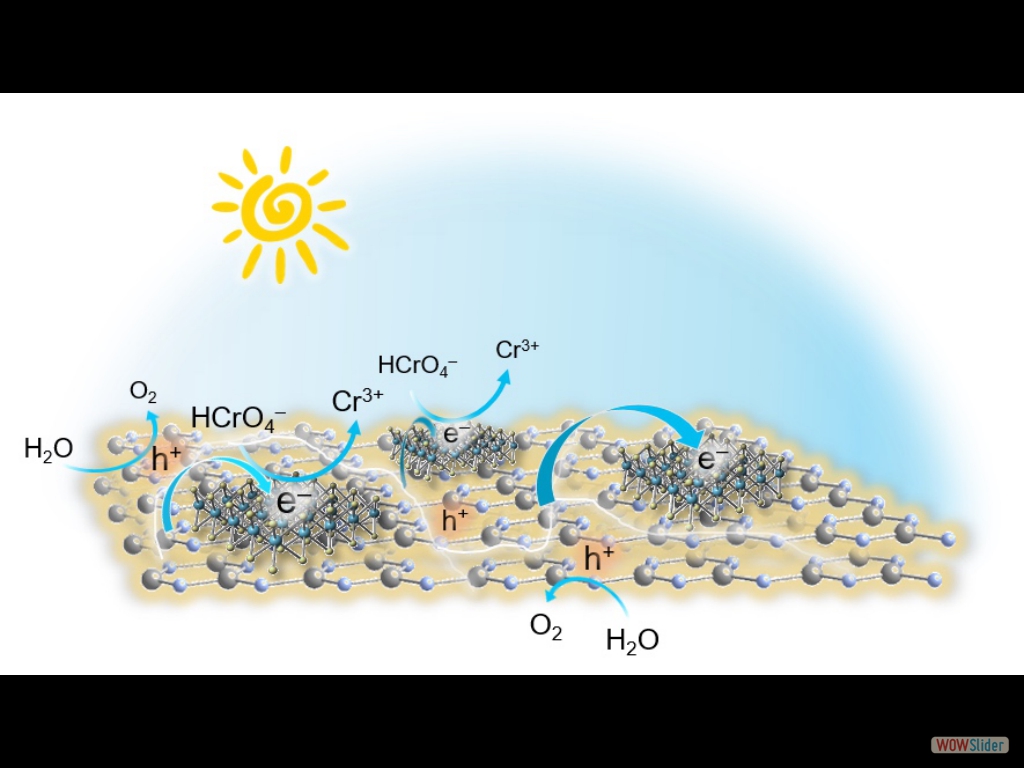 Here, we report that 2D/2D layer heterostructures composed of exfoliated Ni-doped MoS₂ nanosheets and g-C₃N₄ layers can carry out photocatalytic Cr(VI) reduction in aqueous solutions with outstanding activity, exhibiting apparent QYs as high as 29.6 % and 23.7 % at 375 and 410 nm. We show that Ni doping of MoS₂ markedly increases the photochemical activity, which, together with electrochemical, spectroscopic and theoretical DFT studies, arises from the enhanced carrier density and mobility at the Ni-MoS₂/g-C₃N₄ interface.
Here, we report that 2D/2D layer heterostructures composed of exfoliated Ni-doped MoS₂ nanosheets and g-C₃N₄ layers can carry out photocatalytic Cr(VI) reduction in aqueous solutions with outstanding activity, exhibiting apparent QYs as high as 29.6 % and 23.7 % at 375 and 410 nm. We show that Ni doping of MoS₂ markedly increases the photochemical activity, which, together with electrochemical, spectroscopic and theoretical DFT studies, arises from the enhanced carrier density and mobility at the Ni-MoS₂/g-C₃N₄ interface. The Cover Feature shows a 2D/2D nanojunction architecture composed of exfoliated graphitic carbon nitride (g-C₃N₄) flakes and 2H MoS₂ nanosheets. By tuning the lateral dimension of the MoS₂ layers, this system possesses a balanced combination of relatively high electronic communication and large number of catalytic MoS₂ edge sites at the interfacial heterojunctions, thus enabling highly efficient photocatalytic hydrogen production from water. More information can be found in the Full Paper by G. S. Armatas and co-workers (DOI: 10.1002/cplu.201000096).
The Cover Feature shows a 2D/2D nanojunction architecture composed of exfoliated graphitic carbon nitride (g-C₃N₄) flakes and 2H MoS₂ nanosheets. By tuning the lateral dimension of the MoS₂ layers, this system possesses a balanced combination of relatively high electronic communication and large number of catalytic MoS₂ edge sites at the interfacial heterojunctions, thus enabling highly efficient photocatalytic hydrogen production from water. More information can be found in the Full Paper by G. S. Armatas and co-workers (DOI: 10.1002/cplu.201000096).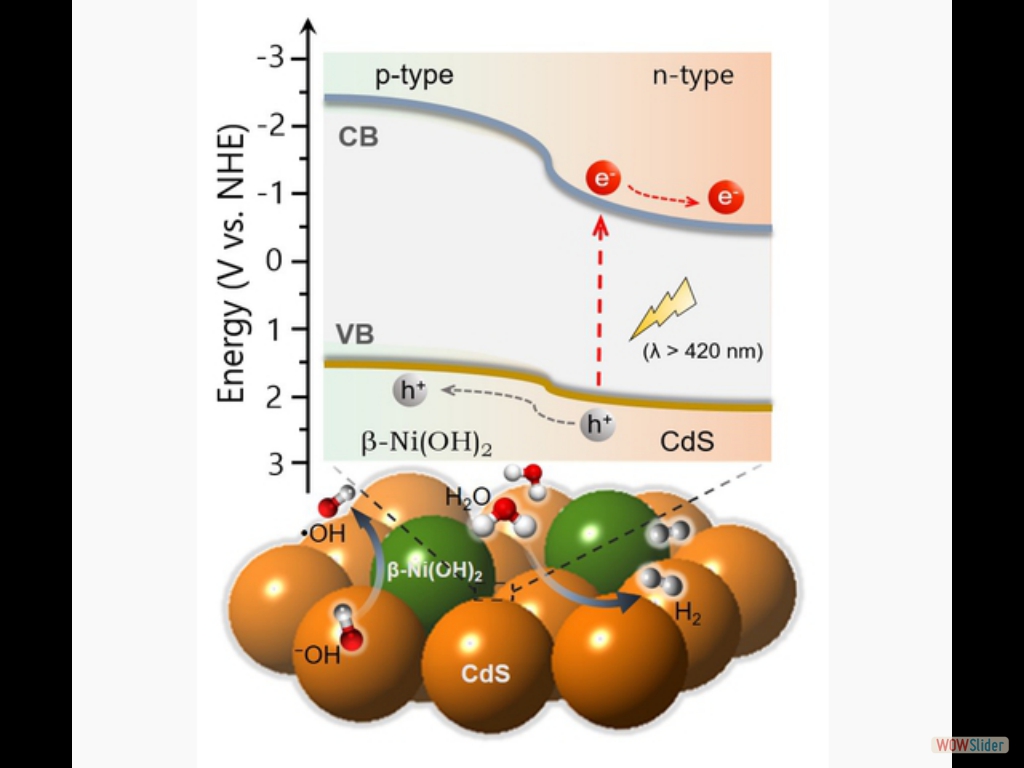 Here, we demonstrate high-surface-area mesoporous networks comprising interconnected β-Ni(OH)₂ modified CdS nanocrystals (NCs) as highly active and stable photocatalysts for hydrogen generation. Compared to single-component CdS assemblies, Ni-modified materials present a strong enhancement of photocatalytic performance for hydrogen evolution under visible light irradiation. Mechanistic study with UV–vis/NIR, PL and EIS spectroscopy and photocatalytic performance evaluation reveals that the improved photocatalytic performance arises from the strong electronic coupling and charge-transferred states at the p–n β-Ni(OH)₂/CdS heterojunctions.
Here, we demonstrate high-surface-area mesoporous networks comprising interconnected β-Ni(OH)₂ modified CdS nanocrystals (NCs) as highly active and stable photocatalysts for hydrogen generation. Compared to single-component CdS assemblies, Ni-modified materials present a strong enhancement of photocatalytic performance for hydrogen evolution under visible light irradiation. Mechanistic study with UV–vis/NIR, PL and EIS spectroscopy and photocatalytic performance evaluation reveals that the improved photocatalytic performance arises from the strong electronic coupling and charge-transferred states at the p–n β-Ni(OH)₂/CdS heterojunctions.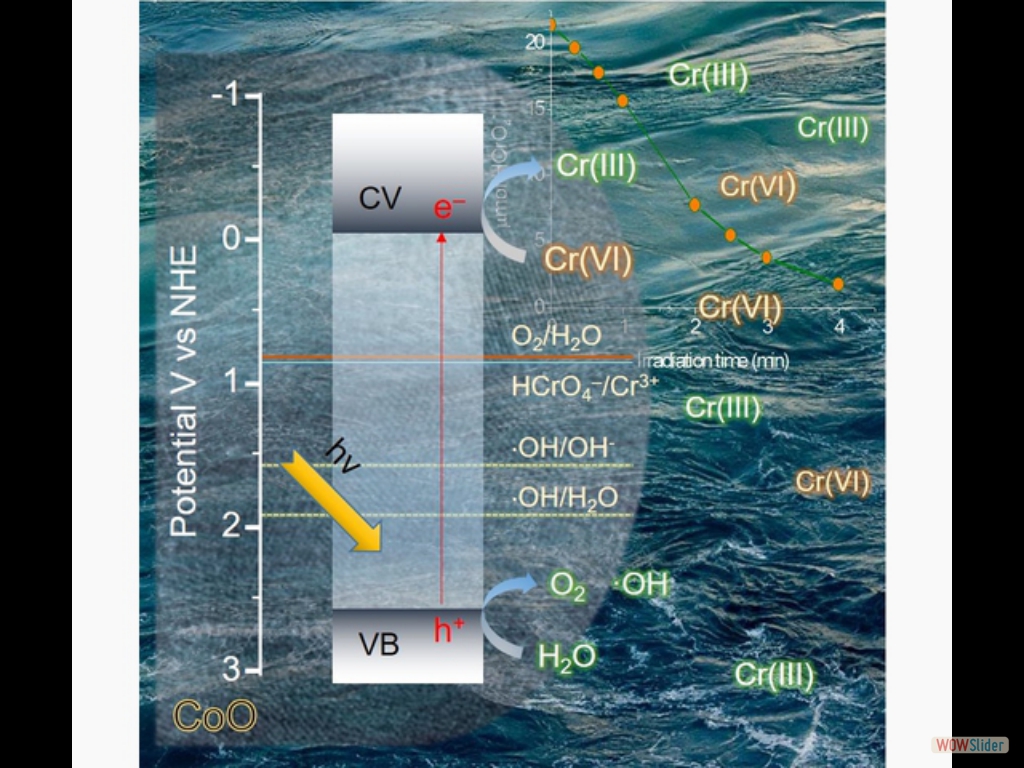 In this work, we report the synthesis of high-surface-area mesoporous networks of CoO NPs through a polymer-templating self-assembly method and demonstrate their potential application in the reductive detoxification of aqueous Cr(VI) solutions under UV and visible light irradiation. We show that mesoporous assemblies of hexagonal CoO NPs effectively overcome the kinetic barriers for the oxidation reaction, manifesting a remarkably photocatalytic Cr(VI) reduction activity at acidic pH with an apparent quantum yield (AQY) of 1.61% and 0.17% at wavelengths of 375 and 440 nm, respectively.
In this work, we report the synthesis of high-surface-area mesoporous networks of CoO NPs through a polymer-templating self-assembly method and demonstrate their potential application in the reductive detoxification of aqueous Cr(VI) solutions under UV and visible light irradiation. We show that mesoporous assemblies of hexagonal CoO NPs effectively overcome the kinetic barriers for the oxidation reaction, manifesting a remarkably photocatalytic Cr(VI) reduction activity at acidic pH with an apparent quantum yield (AQY) of 1.61% and 0.17% at wavelengths of 375 and 440 nm, respectively.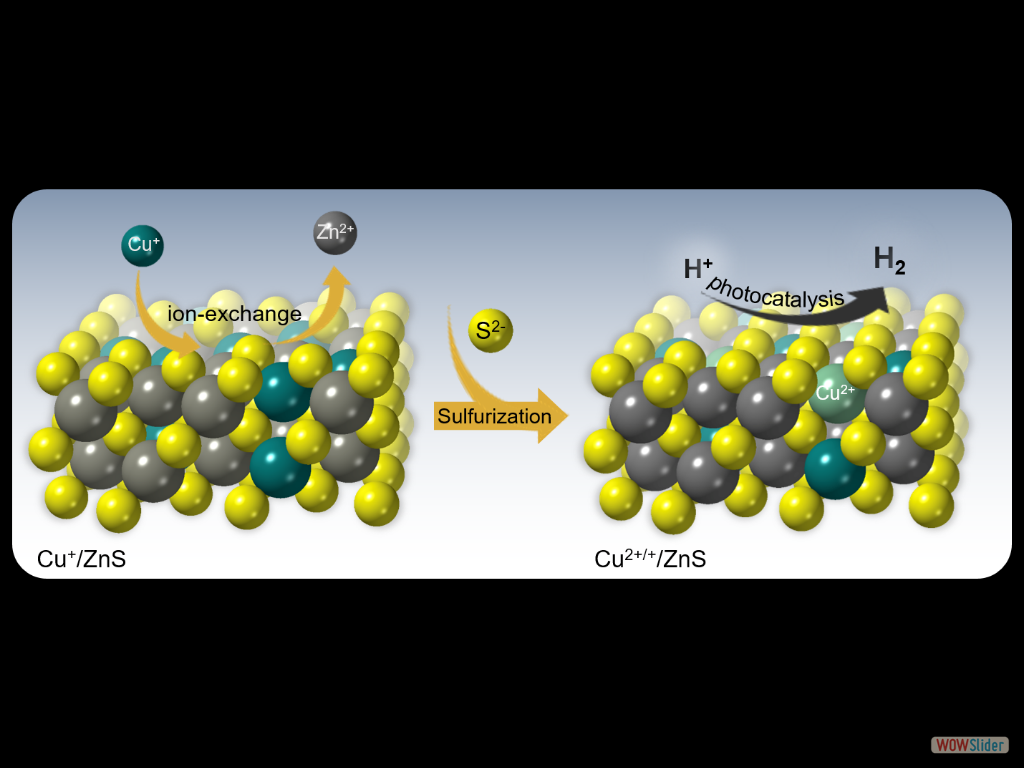 In the present work, high-surface-area Cu-doped ZnS nanocrystal (NC)-linked mesoporous frameworks are successfully synthesized for use as cost-effective catalysts for photochemical hydrogen evolution. Benefiting from the proper band-edge alignment and enhanced visible light absorption resulted from interfacial charge transfer between ZnS and Cu₂S NCs, there is a spatial separation of charge carries which leads to excellent activity for the photocatalytic hydrogen production. Moreover, results here show that surface defect passivation through a wet-chemical sulfidation process effectively increases the photochemical performance of the composite catalysts by improving the transport efficiency of electrons at the Cu₂S/ZnS interface and changing the Helmholtz layer potential drop at the ZnS/Cu₂S/electrolyte junction.
In the present work, high-surface-area Cu-doped ZnS nanocrystal (NC)-linked mesoporous frameworks are successfully synthesized for use as cost-effective catalysts for photochemical hydrogen evolution. Benefiting from the proper band-edge alignment and enhanced visible light absorption resulted from interfacial charge transfer between ZnS and Cu₂S NCs, there is a spatial separation of charge carries which leads to excellent activity for the photocatalytic hydrogen production. Moreover, results here show that surface defect passivation through a wet-chemical sulfidation process effectively increases the photochemical performance of the composite catalysts by improving the transport efficiency of electrons at the Cu₂S/ZnS interface and changing the Helmholtz layer potential drop at the ZnS/Cu₂S/electrolyte junction.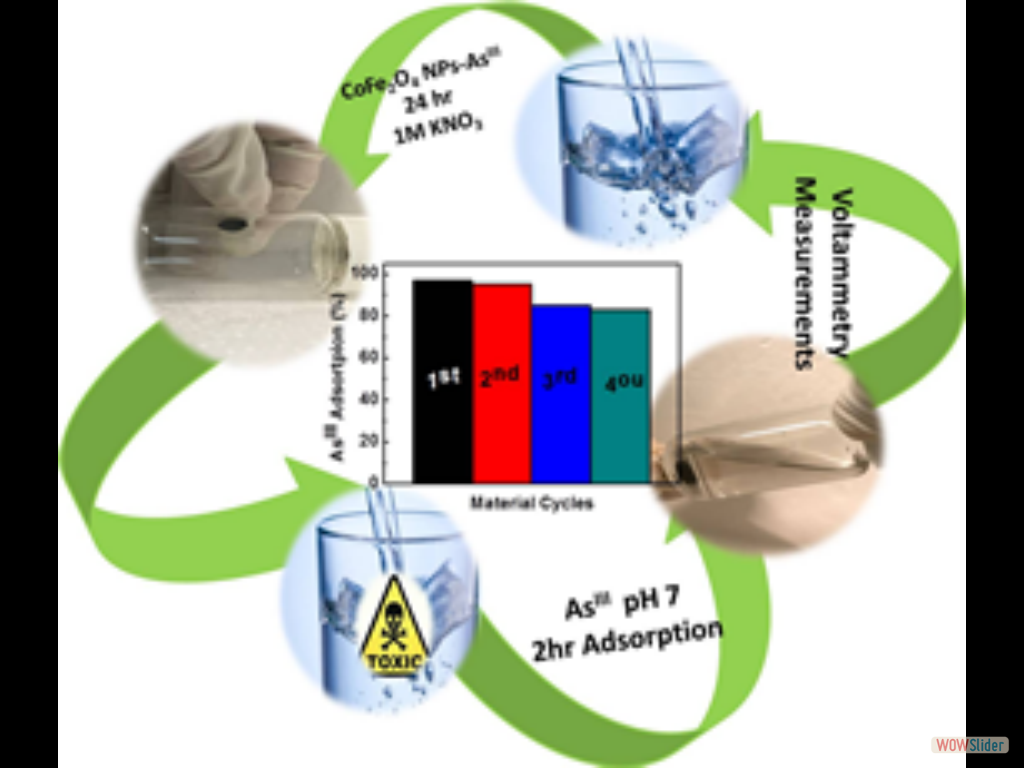 A 3D CoFe₂O₄ mesoporous network of ~6 nm cobalt ferrite (CoFe₂O₄) nanoparticles (NPs), synthesized through a polymer-assisted aggregating self-assembly method, is presented. Owing to its 3D network assemblage, this mesoporous CoFe₂O₄ exhibits an exceptional Asᴵᴵᴵ uptake capacity of 252.8 mg g⁻¹, which is much higher than that of random CoFe₂O₄ NP aggregates (47.3 mg g⁻¹) and bulk-like CoFe₂O₄ microparticles (43.6 mg g⁻¹). A comprehensive surface complexation model is presented, allowing a quantitative description of the Asᴵᴵᴵ adsorption on Fe and Co-sites.
A 3D CoFe₂O₄ mesoporous network of ~6 nm cobalt ferrite (CoFe₂O₄) nanoparticles (NPs), synthesized through a polymer-assisted aggregating self-assembly method, is presented. Owing to its 3D network assemblage, this mesoporous CoFe₂O₄ exhibits an exceptional Asᴵᴵᴵ uptake capacity of 252.8 mg g⁻¹, which is much higher than that of random CoFe₂O₄ NP aggregates (47.3 mg g⁻¹) and bulk-like CoFe₂O₄ microparticles (43.6 mg g⁻¹). A comprehensive surface complexation model is presented, allowing a quantitative description of the Asᴵᴵᴵ adsorption on Fe and Co-sites.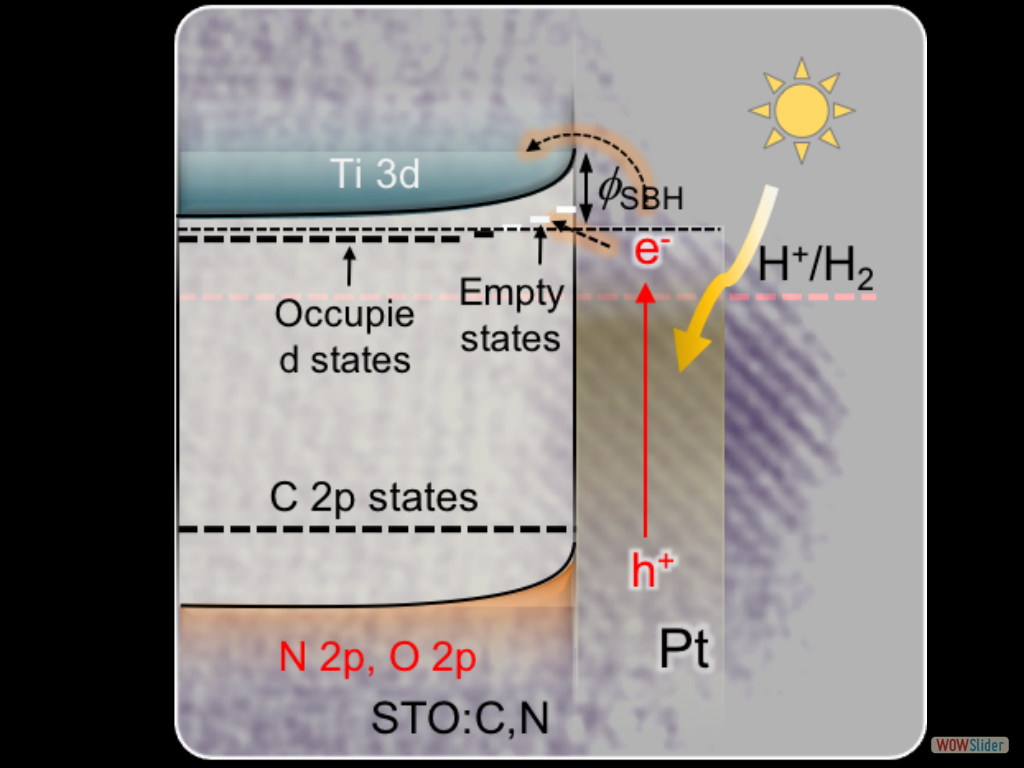 In this study, we devise a facile polymer-assisted sol-gel chemical method to prepare highly porous, crystalline implanted SrTiO₃ (STO) nanoparticles and demonstrate their performance for photocatalytic hydrogen generation from water. Interestingly, a partial substitution of N and C for O is attained in STO lattice with this synthetic protocol, according to the elemental analysis, and infrared (IR) and Xray photoelectron spectroscopy (XPS) studies. Compared to STO:C,N, the STO:C,N mesoporous decorated with Pt nanoparticles (ca. 3 nm) present unique attributes that allow for an impressive improvement of up to 74-fold in photocatalytic H₂-production activity.
In this study, we devise a facile polymer-assisted sol-gel chemical method to prepare highly porous, crystalline implanted SrTiO₃ (STO) nanoparticles and demonstrate their performance for photocatalytic hydrogen generation from water. Interestingly, a partial substitution of N and C for O is attained in STO lattice with this synthetic protocol, according to the elemental analysis, and infrared (IR) and Xray photoelectron spectroscopy (XPS) studies. Compared to STO:C,N, the STO:C,N mesoporous decorated with Pt nanoparticles (ca. 3 nm) present unique attributes that allow for an impressive improvement of up to 74-fold in photocatalytic H₂-production activity.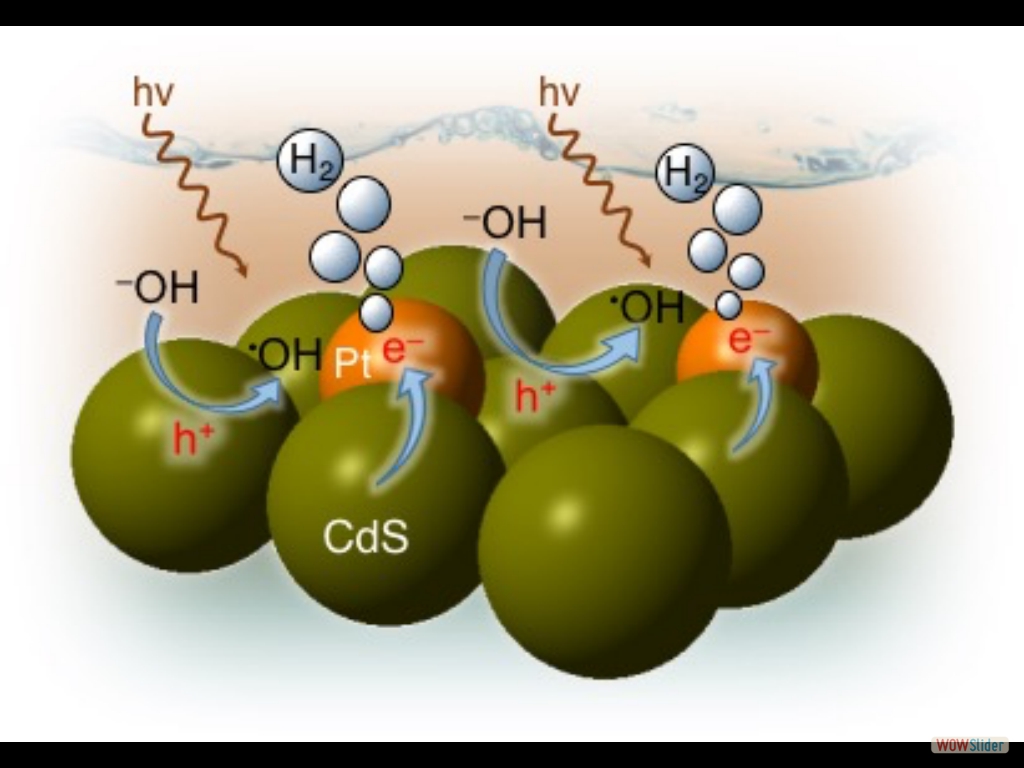 Herein, the design and fabrication of mesoporous Pt‐decorated CdS nanocrystal assemblies (NCAs) are reported, and their excellent performance for the photocatalytic hydrogen production is demonstrated. These materials comprise varying particle size of Pt (ranging from 1.8 to 3.3 nm) and exhibit 3D nanoscale pore structure within the assembled network. Photocatalytic measurements coupled with UV–vis/NIR optical absorption, photoluminescence, and electrochemical impedance spectroscopy studies suggest that the performance enhancement of these catalytic systems arises from the efficient hole transport at the CdS/electrolyte interface and interparticle Pt/CdS electron‐transfer process as a result of the deposition of Pt.
Herein, the design and fabrication of mesoporous Pt‐decorated CdS nanocrystal assemblies (NCAs) are reported, and their excellent performance for the photocatalytic hydrogen production is demonstrated. These materials comprise varying particle size of Pt (ranging from 1.8 to 3.3 nm) and exhibit 3D nanoscale pore structure within the assembled network. Photocatalytic measurements coupled with UV–vis/NIR optical absorption, photoluminescence, and electrochemical impedance spectroscopy studies suggest that the performance enhancement of these catalytic systems arises from the efficient hole transport at the CdS/electrolyte interface and interparticle Pt/CdS electron‐transfer process as a result of the deposition of Pt. The cover picture shows a porous polymeric framework composed of lacunary Keggin‐type [XM11O39]q⁻ (X=P, Si; M=W, Mo) polyoxometalate clusters connected by ethane-bridged silsesquioxane linkers. These new hybrid polymers present a relatively high photocatalytic activity for the hydrogen‐evolution reaction with remarkable cycle stability under UV/visible light irradiation, without any co‐catalysts or additional photosensitizes. Details are given in the Full Paper by G. Armatas et al. on page 947 in Issue 9, 2016 (DOI: 10.1002/cplu.201600199).
The cover picture shows a porous polymeric framework composed of lacunary Keggin‐type [XM11O39]q⁻ (X=P, Si; M=W, Mo) polyoxometalate clusters connected by ethane-bridged silsesquioxane linkers. These new hybrid polymers present a relatively high photocatalytic activity for the hydrogen‐evolution reaction with remarkable cycle stability under UV/visible light irradiation, without any co‐catalysts or additional photosensitizes. Details are given in the Full Paper by G. Armatas et al. on page 947 in Issue 9, 2016 (DOI: 10.1002/cplu.201600199).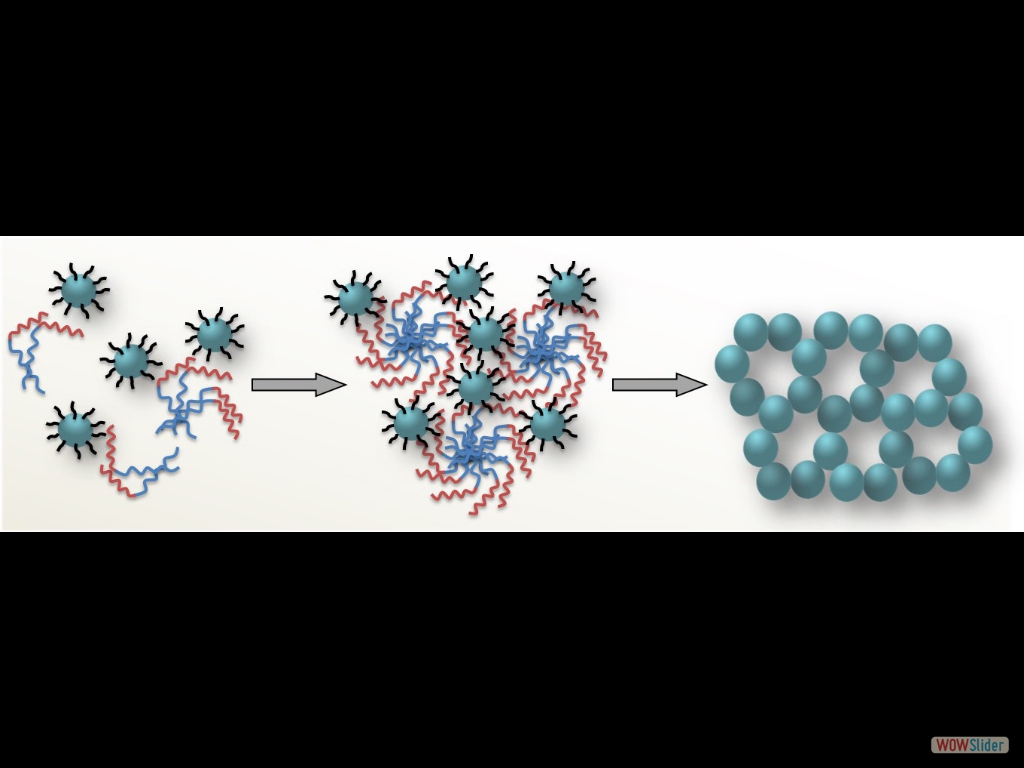 In this perspective, we describe how colloidal nanocrystals can be used as functional building blocks to construct highly porous networks with large and accessible surface area. The synthesis of these mesostructured assemblies, however, is not a simple process and often requires more sophisticated and elegant processing steps. We focused particularly on the potential of a polymer templating technique for the construction of ordered mesostructured assemblies of metal oxide and metal chalcogenide NCs and concentrate on the application of these materials to catalysis.
In this perspective, we describe how colloidal nanocrystals can be used as functional building blocks to construct highly porous networks with large and accessible surface area. The synthesis of these mesostructured assemblies, however, is not a simple process and often requires more sophisticated and elegant processing steps. We focused particularly on the potential of a polymer templating technique for the construction of ordered mesostructured assemblies of metal oxide and metal chalcogenide NCs and concentrate on the application of these materials to catalysis. Mesoporous metal oxides that contain polyoxometalate (POM) compounds are highly attractive materials for a wide range of catalytic applications. The incorporation of POM components within a porous inorganic support may endow intriguing functionalities to the parent materials such as high catalytic activity and recyclability. A substantial progress has been made in the synthesis of new mesoporous polyoxometalate-based catalysts by employing post-synthesis grafting or wet impregnation of POM units onto inorganic supports or direct sol-gel reaction of metal oxide species and POM compounds. This chapter highlights the recent achievements in this area emphasizing the preparation and catalytic properties of these materials.
Mesoporous metal oxides that contain polyoxometalate (POM) compounds are highly attractive materials for a wide range of catalytic applications. The incorporation of POM components within a porous inorganic support may endow intriguing functionalities to the parent materials such as high catalytic activity and recyclability. A substantial progress has been made in the synthesis of new mesoporous polyoxometalate-based catalysts by employing post-synthesis grafting or wet impregnation of POM units onto inorganic supports or direct sol-gel reaction of metal oxide species and POM compounds. This chapter highlights the recent achievements in this area emphasizing the preparation and catalytic properties of these materials.
Current research activities focused on design, synthesis and characterization of nanostructured materials and their applications, including:
Links to the articles are provided. The abstract and article information are accessible, however, subscription to the journal requires for the full text.
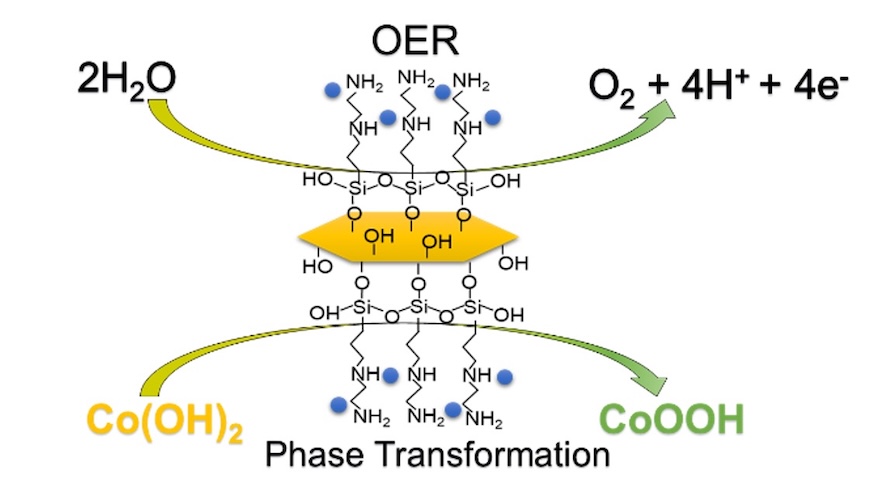
[151] N. Moscholakis, M.S. Metaxa, E.D. Papadaki, G. Potsi, G.S. Armatas & A. Kouloumpis. Surface-Engineered α-ZrP Nanoplatelets as a Platform for Cobalt-based Oxygen Evolution Electrocatalysis. ACS Appl. Nano Mater., 9, 9863–9874 (2026).
In this study, we report a surface-engineering strategy that transforms layered α-zirconium phosphate (α-ZrP) nanoplatelets into an expanded and chemically robust two-dimensional (2D) platform for the oxygen evolution reaction (OER). Covalent grafting with diamino organosilanes introduces pendant amine functionalities that serve as well-defined coordination sites, enabling strong immobilization and controlled nucleation of cobalt species. Structural and spectroscopic analyses reveal substantial interlayer expansion together with the preferential formation of a surface-confined Co(OH)₂ phase on the functionalized α-ZrP framework. Under alkaline OER conditions, the catalyst undergoes in situ electrochemical reconstruction to form catalytically active CoOOH species, which constitute the operative phase during water oxidation. The resulting Co-am-ZrP electrocatalyst delivers low overpotentials of 278 and 327 mV at current densities of 10 and 50 mA cm-2 respectively, together with a Tafel slope of 64.7 mV dec-1, indicative of favorable OER kinetics. Moreover, the catalyst maintains stable electrocatalytic activity over 50 h of continuous operation without significant performance loss. This work establishes functionalized α-ZrP nanoplatelets as versatile and robust 2D scaffolds for the rational design of earth-abundant electrocatalysts and highlights the critical role of synergistic ligand–metal–support interactions within covalently engineered layered architectures in regulating cobalt speciation and electrochemical reconstruction behavior.
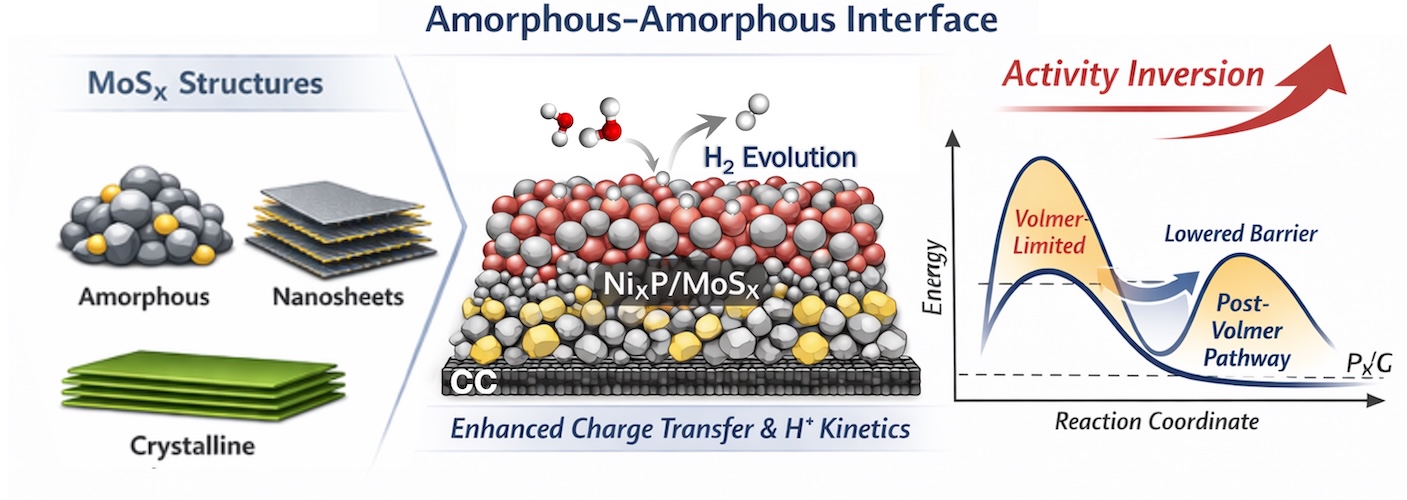
[150] I. Vamvasakis, A. Grammenos & G.S. Armatas. Reversing the Activity Landscape of MoSx Electrocatalysts via NixP Interfacial Coupling for Alkaline Hydrogen Evolution. Chem. Commun., in press (2026).
Interfacial coupling offers a powerful route to modulate electrochemical kinetics, yet its interplay with catalyst morphology remains unclear. Here, NixP coupling reshapes the activity landscape of MoSx, enabling a highly active amorphous interface for alkaline HER. Operando electrochemical analysis reveals a shift beyond Volmer-limited kinetics via enhanced charge transfer and hydrogen adsorption.
(*This article is part of the themed collection: ChemComm Electrocatalysis)
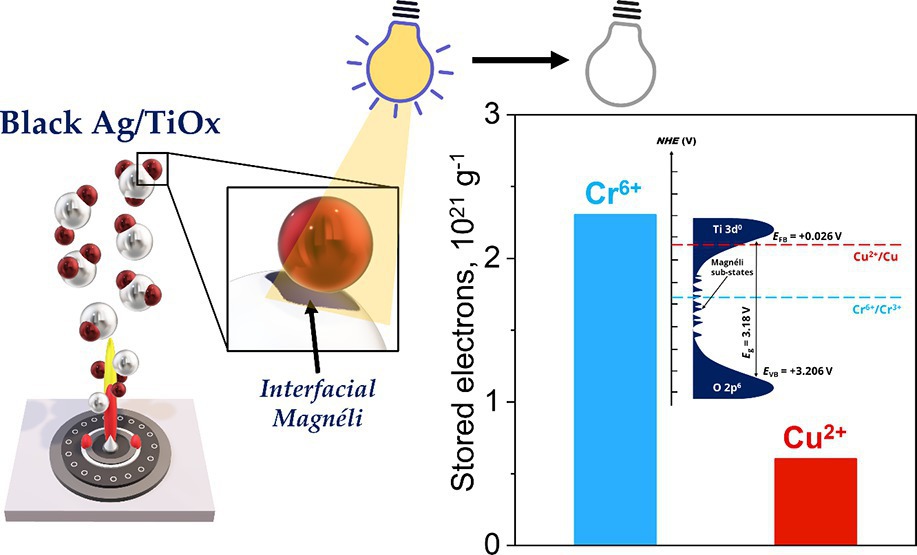
[149] C. Moularas, C. Dimitriou, S. Smykała, I. Vamvasakis, G.S. Armatas & Y. Deligiannakis. Dark Photocatalysis via Long-Term Storage of Photoinduced Electrons in Black-TiOx/Ag0 Heterojunctions. ACS Appl. Energy Mater., 9, 6037–6049 (2026).
Dark photocatalysts allow storage of photogenerated electrons and their post-irradiation use under no-light conditions. This requires the development of materials able to perform photogeneration of electrons, long-term in situ charge storage, and controlled transfer to acceptors on demand. Herein, we present {Black-TiOx/Ag0} nanohybrids consisting of {Black-TiOx} decorated with plasmonic silver Ag0 nanoparticles produced by flame spray pyrolysis (FSP). A library of {Black-TiOx/Ag0} nanocomposites was engineered, with precise control over Ag0 loading and {Black-TiOx} particle size. Under dark photocatalysis conditions, pre-irradiated {Black-TiOx/Ag0} exhibits significant capacity to store and transfer electrons to Cr6+ or Cu2+ ions under dark conditions. Specifically, pre-irradiated {Black-TiOx/Ag0} provides 0.6 × and 2.3 × 1021 electrons g-1 of {Black-TiOx/Ag0} to Cr6+ ions and Cu2+ ions, respectively, for at least 120 min. Operando electron paramagnetic resonance (EPR) spectroscopy reveals that Magneli-TiOx surface sites on {Black-TiOx/Ag0} act as efficient trap/store units of the photoexcited electrons. The interfacial Magnéli islands introduce sub-gap energy states in TiO2, drastically improving both electron storage and visible-light activity compared to conventional TiO2/Ag0. Photoluminescence and photoelectrochemical analyses show that {Black-TiOx/Ag0} exhibits high electron mobility and low hole/electron recombination rates. This analysis reveals that the interfacial Magnéli islands in contact with Ag0 act [i] as efficient electron storage pools of surface electrons under sunlight and [ii] as long-lived electron donors in the dark. This work provides a first proof-of-concept for {Black-TiOx/Ag0} nanocomposites as photochargeable materials, enabling decoupled dark redox chemistry, while highlighting FSP as a versatile and scalable route for their synthesis.
[148] E.-O. Alexandridou, I.V. Vasiliadou, T. Ioannidou, E. Kalpakidou, V. Keller, D. Papoulis, I.T. Papadas, G.S. Armatas & K.C. Christoforidis. Clay/MOF composites for cationic/anionic dyes and CO2 adsorption. Int. J. Environ. Anal. Chem., 1–20 (2026).
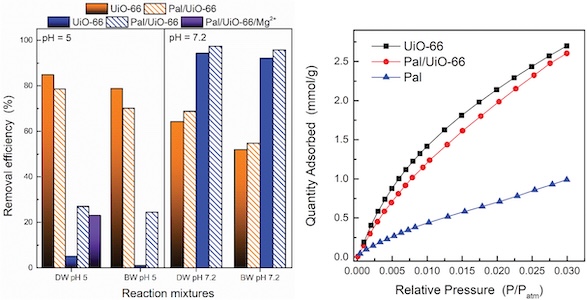
Herein, we present the synthesis of a novel clay (palygorskite (Pal)) functionalised metal-organic framework (MOF)-based (UiO-66) composite applied as adsorbent for water and air treatment. The efficiency for the removal of both cationic and anionic dyes from aqueous matrices and the CO2 capture was evaluated. The composite was synthesised via an easy open-vessel thermal process. The structural, textural, compositional and morphological properties of the prepared materials were characterised using a variety of techniques including X-ray diffraction (XRD), attenuated total reflection (ATR), N2 physisorption, thermogravimetric analysis (TGA), scanning electron microscopy (SEM) and X-ray photoelectron spectroscopy (XPS). The adsorption properties of the materials were evaluated for the removal of methylene blue (MB) and methyl orange (MO), as representative cationic and anionic dyes, respectively. Their efficiency in CO2 capture efficiency was also investigated. All UiO-66-based materials presented high efficiency for the removal of MO. Despite that Pal showed very low MO removal efficiency, its presence did not alter the adsorption of MO on the Pal/UiO-66 composite. On the contrary, the Pal/UiO-66 was much more efficient than pristine UiO-66 for the removal of cationic dyes. This is ascribed to the presence of palygorskite, offering multifunctionality for the treatment of waters containing both anionic and cationic dyes. In addition, although Pal presented low CO2 uptake ability, its presence in the composite did not alter significantly the CO2 adsorption efficiency of the composite compared with the pristine MOF. The findings of this study demonstrate the significance of coupling clays with MOF structures for the development of synergies that allow multifunctionality, expanding the application of MOF-based sorbents.
[147] A.G. Margellou, G.S. Armatas, K. Kolonia & P.J. Pomonis. Correlation Between Information Entropy and Pore Connectivity in Oxidic Materials with Random Disordered Porosity. Phys. Chem. Chem. Phys., 28, 2574–2584 (2026).
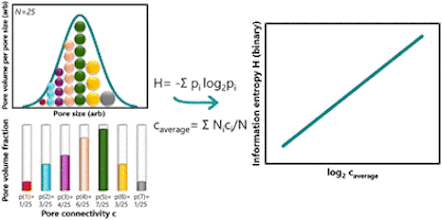
Pore connectivity is a crucial structural characteristic of porous solids that governs the mass transfer and diffusion of fluids through them. Information entropy, on the other hand, is a statistical property that can be estimated for any distribution, including the size distribution of pores in solids. In this work, we present the first comprehensive study investigating the correlation between the pore connectivity and the Shannon information entropy of classical pore size distributions of inorganic porous materials. Experimental data are based on sixteen reported oxidic alumino–phosphoro–vanadate porous solids. All studied materials exhibit random disordered porosity, as determined by standard nitrogen porosimetry. Pore connectivity was estimated using the Seaton method, which is based on the hysteresis loop of nitrogen adsorption–desorption isotherms. Results demonstrate a linear and robust correlation between the binary information entropy and binary logarithm of pore connectivity. This relationship can be rationalized by considering the statistical information entropy of independent pore mixing. The physical origin of the correlation between information entropy and average pore connectivity is attributed to the random packing of pores, analogous to the classical problem of random packing of particles. The statistical base is the heteroscedasticity between the variance and mean parameters of pore size distributions: variance drives information entropy, while the mean drives pore connectivity, as described by the model of random packing.
[146] M.S. Metaxa, I. Vamvasakis & G.S. Armatas. Heterointerface Engineering of FeOOH@Ni3N Electrocatalysts for Industrially Compatible Alkaline Water Electrolysis. Small, 22, e13136 (2026).
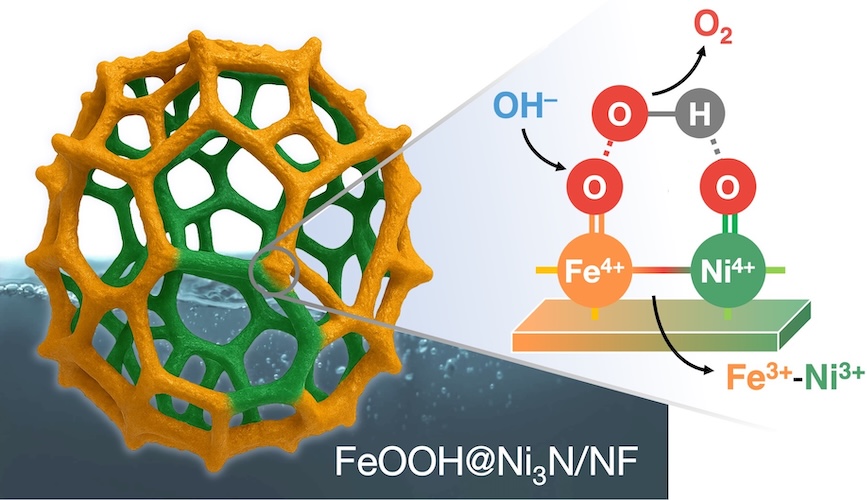
The rational design of earth-abundant electrocatalysts is pivotal for advancing alkaline water electrolysis toward sustainable hydrogen production. Here, we report a hierarchical FeOOH@Ni3N heterostructure comprising a redox-active iron oxyhydroxide overlayer conformally coupled with a conductive trinickel nitride core directly grown on nickel foam. This hybrid catalyst drives the oxygen evolution reaction (OER) with ultralow overpotentials of 209, 245 and 284 mV at 10, 100, and 500 mA cm⁻2, respectively, while maintaining exceptional stability under industrial-level operations. Integrated into a two-electrode electrolyzer, FeOOH@Ni3N achieves current densities of 10, 500 and 1000 mA cm⁻2 at cell voltages of only 1.49, 1.72 and 1.78 V, outperforming noble-metal-based benchmarks. Operando/in-situ spectroscopies, combined with electrokinetic and isotope-effect analyses, reveal that enhanced intrinsic activity originates from reconstructed proton–electron transfer pathways at the Fe–Ni heterointerface. Strong interfacial coupling stabilizes high-valent Ni4+=O and Fe4+=O species and promotes an unconventional dual-site hydroxyl nucleophilic attack (HNA) mechanism, wherein OH⁻ attack on Fe4+=O forms a bridging *OOH intermediate as the O–O bond-forming step, synergistically assisted by adjacent Ni centers. These findings delineate a clear structure–activity–stability relationship for Fe–Ni heterostructures and showcase heterointerface engineering of conductive nitrides with oxyhydroxides as a scalable strategy for developing durable, high-rate OER electrocatalysts.
[145] M.S. Metaxa, I. Vamvasakis & G.S. Armatas. Rational Design and Operando Characterization of Hierarchical α-FeOOH-FeP/Ni3S2 Catalysts for High-Rate Alkaline Water Electrolysis. Energy Environ. Mater., 9, e70187 (2026).
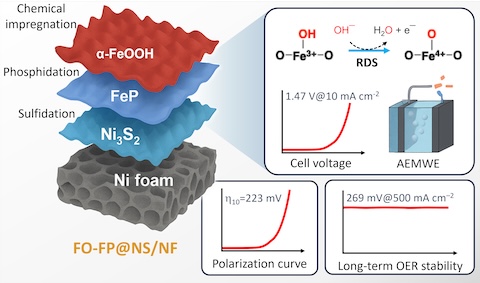
Advancing alkaline water electrolysis for renewable energy technologies requires oxygen evolution reaction (OER) electrocatalysts that combine high activity, long-term durability and mechanistic clarity. Herein, we report a hierarchically engineered α-FeOOH–FeP/Ni3S2 electrocatalyst supported on 3D Ni foam, synthesized via a stepwise hydrothermal sulfidation, gas-phase phosphidation, and chemical impregnation strategy. This integrated multi-phase architecture exhibits strong interfacial coupling, enabling accelerated charge transfer and favorable OER kinetics under alkaline conditions. In-situ/operando Raman, UV-vis, and electrochemical impedance spectroscopy uncover dynamic surface reconstruction under operating conditions, with reversible Fe3+/Fe4+ redox cycling within the α-FeOOH overlayer, pinpointing transient Fe4+–O species as key catalytic intermediates. The optimized catalyst attains low overpotentials of 223 and 251 mV at 10 and 100 mA cm-2 and sustains industrial-level operation (> 500 mA cm-2) with outstanding durability in 1.0 M KOH. When deployed in a symmetric anion exchange membrane water electrolyzer, it delivers a cell voltage of only 1.47 V at 10 mA cm–2, outperforming benchmark noble-metal-based systems. Mechanistic studies including kinetic isotope effect and pH-dependent analysis support a proton-coupled electron transfer (PCET) mechanism, with O–H bond cleavage as the rate-determining step. These findings elucidate key structure-function relationships and establish a modular design strategy for advanced alkaline OER electrocatalysts.
(*This article belongs to the Special Issue: Sustainable multi‐functional materials for energy and environmental application)
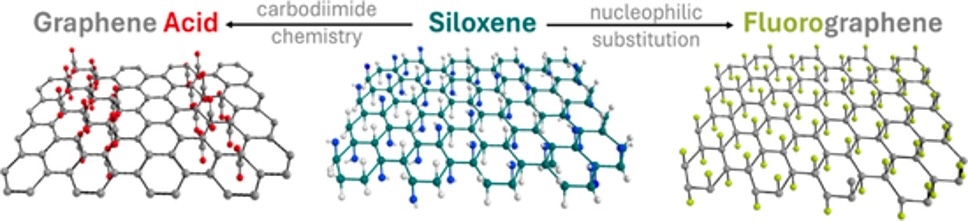
[144] N. Moscholakis, G. Potsi, D.D. Chronopoulos, I. Vamvasakis, G.S. Armatas & A. Kouloumpis. Covalent Integration of Siloxene and Graphene Derivatives into 2D Heterostructures as Metal-Free Catalysts for the Oxygen Evolution Reaction. Appl. Mater. Today, 47, 102987 (2025).
While physically stacked 2D heterostructures offer synergistic properties, their reliance on weak van der Waals interactions often limits long-term stability. In contrast, covalently bonded 2D heterostructures provide durable interfaces and precisely controlled interlayer spacing, unlocking more effective synergistic effects. Herein, we report the synthesis of robust 2D/2D hybrids by covalently linking amine-functionalized siloxene nanosheets (fSN) with fluorographene (FG) via nucleophilic substitution and with graphene acid (GA) via carbodiimide-mediated amide coupling. Structural and spectroscopic analyses confirmed the formation of stable C–N and amide linkages, alongside the preservation of the constituent 2D architectures. As metal-free oxygen evolution reaction (OER) catalysts in alkaline media, FG–fSN achieves an overpotential of 370 mV at 10 mA cm⁻², outperforming the individual components due to accelerated charge transfer, increased electrochemically active surface area, and enhanced stability. GA–fSN also demonstrates improved OER activity and durability, though to a lesser extent, underscoring the critical role of interfacial chemistry. These results establish covalent siloxene–graphene derivative heterostructures as a versatile and high-performance platform for durable metal-free electrocatalysts, offering a promising route for next-generation energy conversion technologies.
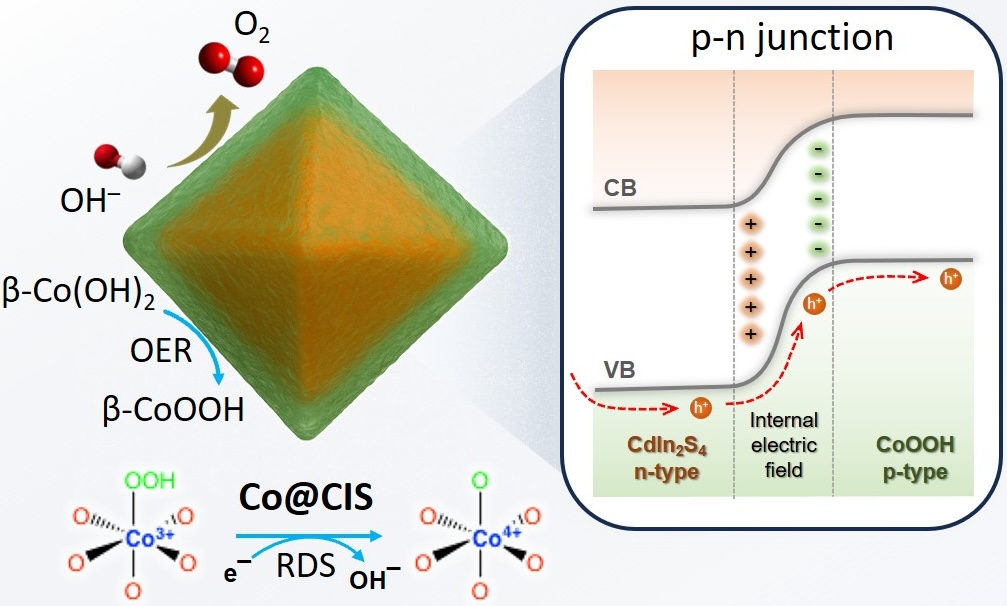
[143] M.S. Metaxa, I. Vamvasakis, G. Kopidakis, G. Kioseoglou & G.S. Armatas. Interfacial Engineering of p-n β-Co(OH)2/CdIn2S4 Heterojunctions for Efficient Alkaline Oxygen Evolution Electrocatalysis. Chem. Eng. J., 525, 170118 (2025).
Semiconductor-based electrocatalysts with tunable electronic structures and efficient interfacial charge transfer hold great promise for sustainable energy conversion. In this study, we present a novel nanostructured electrocatalyst composed of β-Co(OH)2 nanolayers uniformly anchored on CdIn2S4 (CIS) thiospinel nanoparticles, synthesized via a combined hydrothermal and photochemical deposition strategy. Comprehensive structural and spectroscopic analyses, supported by theoretical calculations, demonstrate the formation of a well-defined p–n heterojunction at the β-Co(OH)2/CIS interface, which promotes directional charge transport and reduces interfacial resistance, collectively accelerating oxygen evolution reaction (OER) kinetics. The improved electrocatalytic activity arises from the synergistic effects of interfacial electric field modulation, topotactic electrochemical transformation of β-Co(OH)2 into catalytically active β-CoOOH, and stabilization of high-valent Co4+ species under anodic polarization. The optimized 10 wt% Co@CIS catalyst achieves a low overpotential of 294 mV at 10 mA cm–2, a small Tafel slope of 61.2 mV dec–1, and excellent operation stability over 100 h in alkaline electrolyte. When integrated into a two-electrode alkaline water electrolyzer, it enables overall water splitting at a cell voltage of only 1.60 V at 10 mA cm–2. These results highlight the critical importance of interfacial engineering in designing high-performance, durable and cost-effective electrocatalysts for renewable energy conversion technologies.
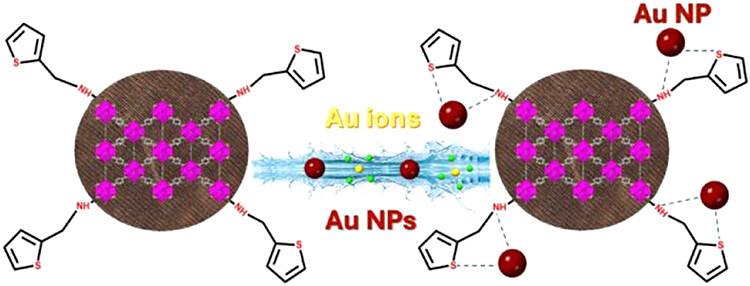
[142] V. Gouma, E.C. Makri, E.K. Andreou, E. Buchsteiner, G.S. Armatas, M.J. Manos & D.L. Giokas. An Amino-Thiophene Functionalized Metal–Organic Framework on Fabric for Selective Extraction, Recovery, and Passive Sampling of Gold Ions and Nanoparticles. Chem. Mater., 37, 7147–7158 (2025).
The steadily increasing use of gold and the limited gold reserves have instigated a significant research effort to develop materials with efficient gold recovery. However, most works focus only on the uptake of Au ions, ignoring Au nanoparticles, which are progressively used in various applications and represent a critical Au resource. Furthermore, the reported gold recovery studies involve powder-form materials that cannot be easily retrieved after the sorption process, thus making them unattractive to real, large-scale applications. The present work reports a new Zr(IV) metal-organic framework (MOF) with a defective microporous structure and amino-thiophene functional groups, which exhibit strong interactions with Au species. The MOF was immobilized on cotton fabric via an in-situ method to create an easily retrievable bulk sorbent composite. It was investigated in detail for its sorption properties towards Au ions and Au nanoparticles (AuNPs). The immobilized sorbent showed relatively fast sorption kinetics (<1 and 3 h for Au ions and AuNPs, respectively), impressive sorption capacities (883.5 and 43.4 mg Au/g for Au ions and AuNPs, respectively), capability for capturing AuNPs irrespective of their coating and size and high sorption efficiency and recovery (>85%) of gold either as ions or NPs from genuine water samples. The latter property allowed the utilization of the MOF-fabric sorbent as the first passive sampling device for the long-term monitoring or recovery of AuNPs from natural waters. Notably, the sorbent was highly effective for the selective Au recovery (~97%) from electronic waste under flow conditions. Not the least, the material could be regenerated and reused for gold capture at least three times.
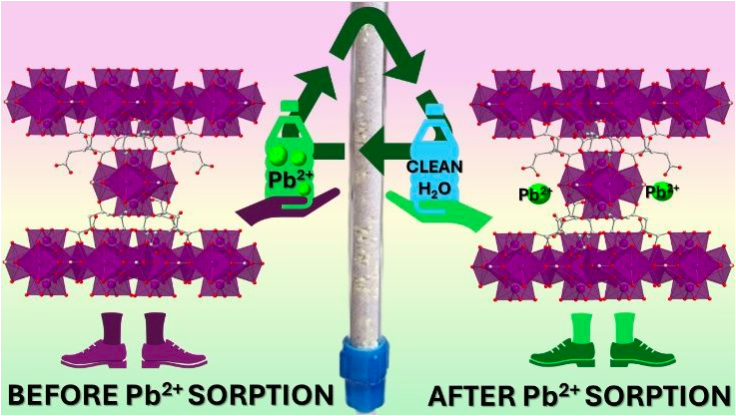
[141] N.E. Anastasiadis, V.I. Karagianni, P. Kotidis, C. Dimitriou, I. Vamvasakis, T. Lazarides, G.S. Papaefstathiou, Y. Deligiannakis, G.S. Armatas, E. Buchsteiner & M.J. Manos. Aqueously Synthesized and Reusable Ce(IV)-Based Metal–Organic Frameworks with Aliphatic Multicarboxylate Linkers for Highly Efficient Pb2+ Removal from Water. Inorg. Chem. Front., 12, 7894-7906 (2025).
New Ce(IV) metal organic frameworks (MOFs) are reported based on the aliphatic linker butane-1,2,3,4-tetracarboxylic acid (H4BTCA). These materials were prepared using an eco-friendly aqueous-based synthesis, and their structures were elucidated through microcrystal electron diffraction (MicroED) and powder X-ray diffraction (PXRD). They feature an 8-coordinated net with a bcu topology, rarely observed for Ce(IV) MOFs. Interconversion between the MOFs was easily achieved via treatment with basic or formic acid solutions, revealing the breathing of the MOFs’ frameworks. The MOFs were investigated for their ability to sorb Pb2+ under both batch and continuous flow conditions. The results showed relatively high Pb2+ sorption capacities (up to 254 mg Pb/g) and an exceptional capability for the rapid removal (in less than 10 min of MOF-solution contact) of Pb2+ from low initial concentrations from complex solutions and real-world water samples. Notably, the materials in the form of calcium alginate-based beads used as a stationary phase in a column (along with sea sand) demonstrate a remarkable ability to capture Pb2+ under continuous flow, showing decent removal capacities, excellent regeneration efficiency, and reusability. The mechanism of the Pb2+ sorption process was determined via experimental data, indicating strong interactions of the MOF’s functional groups (carboxylate and hydroxide groups) with the Pb2+ ions.
[140] D.A. Evangelou, E.C. Makri, N. Pliatsios, I. Vamvasakis, E. Buchsteiner, P. Oikonomopoulos, G.S. Armatas, G.S. Papaefstathiou, T. Lazarides & M.J. Manos. Ultramicroporous Αl(III) MOFs with Selective CO2 Adsorption, Acid Resistance and Efficient Cr(VI) Sorption Properties. Dalton Trans., 54, 13658-13671 (2025).
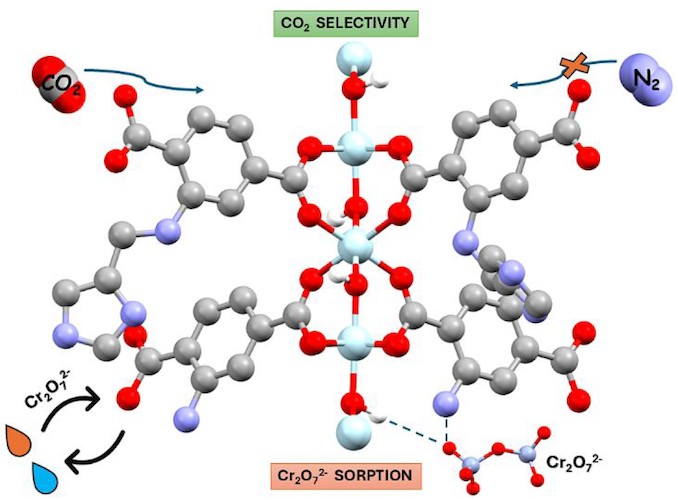
In the present work, we report the hydrothermal synthesis and detailed characterization of a series of ultramicroporous, luminescent, multivariate metal-organic frameworks (MOFs) with the general formula [Al(OH)(IATP)1-x(NH2-BDC)x], constructed from two different dicarboxylate ligands, H2IATP = 2-(((1H-imidazol-4-yl)methyl)amino) terephthalic acid and NH2-H2BDC = 2-aminoterephthalic acid. Structural analysis, using both powder X-ray diffraction (PXRD) and microcrystal electron diffraction (Micro-ED), confirms that the reported MTV-Al-MOFs are topological analogues of the related NH2-MIL-53(Al) MOF. Introducing ultramicroporosity (~5.2 Å) together with bulky imidazole functional groups significantly enhances the chemical stability of these MOFs under acidic conditions, compared to NH2-MIL-53(Al). It also imparts interesting carbon dioxide physisorption properties, with MTV-Al-MOF-4 (Al(OH)(IATP)0.39(NH2-BDC)0.61) demonstrating a maximum CO2 adsorption capacity of 2.33 mmol/g at 273 K and a selectivity CO2/N2 of ~76 at the same temperature (273 K) and low pressure limit. Among the series, MTV-Al-MOF-4 exhibits the most promising combination of chemical stability and Cr(VI) sorption capacity. This material was extensively studied for eliminating Cr2O72- anions from acidic aqueous solutions, indicating high sorption capacity (174 mg Cr2O72-/g), rapid uptake kinetics (equilibrium reached within 5 minutes), and decent selectivity against competing anions such as Cl-, NO3-, and SO42-. For practical applications in wastewater treatment, a MOF calcium alginate (CA) composite material was fabricated in beads and effectively removed Cr(VI) from diluted electroplating waste under dynamic flow conditions. Finally, all MTV-Al-MOFs displayed fluorescence with quantum yields in the range of 4-6%, while Cr(VI)-loaded MTV-Al-MOF-4 shows a pronounced reduction in fluorescence quantum yield by a factor of four, thereby confirming the presence of ligand-sorbent interactions leading to photoinduced energy and/or electron transfer processes. Overall, this study highlights a versatile approach to designing multifunctional MOFs with potential applications in environmental remediation, industrial wastewater treatment, luminescence, and gas separation.
[139] E. Skliri, A.P. Souri, I. Vamvasakis, E. Andreou, G.S. Armatas & V.D. Binas. p‐Cu(OH)2/n‐SrTiO3 Heterojunction for Efficient Photocatalytic Hydrogen Production. Adv. Sustainable Syst., 9, e00127 (2025).
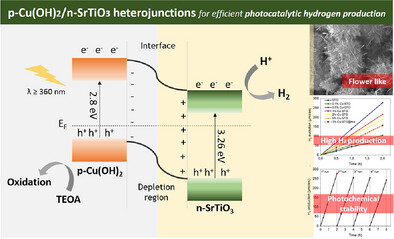
Improving the efficiency of well-known photocatalysts in the hydrogen evolution reaction is contingent upon the development of cost-effective and highly efficient co-catalysts. The rapid recombination of photogenerated charges within semiconductor materials, such as SrTiO3 (STO), results in reduced photocatalytic efficiency. To address this, copper-based co-catalysts have garnered considerable attention due to their relative low-cost, natural abundance of constituents, and promising reactivity. In this work, p-Cu(OH)2/n-SrTiO3 heterojunctions are prepared for efficient photocatalytic hydrogen production. The SrTiO₃ photocatalysts with a flower-like morphology decorated with Cu(OH)2 at different weight percentages of Cu (0.1, 0.5, 1, 2, and 5 wt.%) are successfully prepared using a simple solvothermal and photodeposition process. The p-Cu(OΗ)2/n-SrTiO3 composite with 1 wt.% Cu content exhibits significant enhancement toward photocatalytic hydrogen production compared to the pristine STO, achieving a rate of ∼139 µmol h− 1 (∼6950 µmol g− 1 h− 1 mass activity) under λ > 360 nm light irradiation which is ≈3.5 times higher than that of single-component STO. X-ray photoelectron spectroscopic studies indicate formation of Cu(OH)2 phase. Moreover, UV–vis/NIR, EIS spectroscopy, and photocatalytic evaluation studies indicate that the improved photocatalytic performance arises from the formation of p-n junction at the Cu(OH)2/STO interface that promotes enhanced charge carrier transfer and separation within the heterojunctions.
[138] I.T. Papadas, S.M. Pozov, I. Hamilton, M. Sims, I. Vamvasakis, A. Ioakeimidis, G.S. Armatas, D.D.C. Bradley & S.A. Choulis. Low-Temperature Solution Combustion-Synthesized CuS Nanoparticulated Functional Thin Films: Structural and Optoelectronic Characterization Studies. Nanoenergy Adv., 5, 3 (2025).
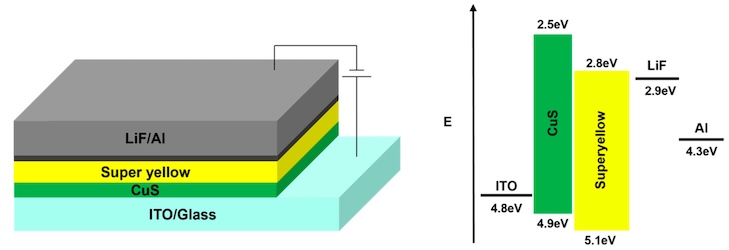
In this paper, we present a one-step low-temperature solution combustion synthesis (SCS) of CuS nanoparticulated functional films processed via a simple blade-coating technique. This SCS route uses thiourea as a fuel and sulfur source, combined with copper(II) nitrate as an oxidant and a cupric ion source in an aprotic solvent such as non-toxic DMSO. It is hereby shown that the proposed SCS process formed a stable and completely dissolved molecular ink of thiourea and copper ion complexes, crucial for obtaining the pure crystalline phase of CuS nanoparticles (NPs). The CuS was formed by calcination at a low temperature of 200 °C during a brief annealing time of 20 min, to promote the synthesis of ~10 nm CuS NPs. The obtained CuS NPs were thoroughly analyzed in terms of structure and optoelectronic properties using various analytic and spectroscopic techniques, including TGA, XRD, FE-SEM, EDS, AFM, and four-point probe electrical resistivity measurements. The functionality of the prepared CuS nanoparticulated interlayers was evaluated by incorporating them as a hole injection layer (HIL) in Super Yellow (SY) organic light-emitting diodes (OLEDs).
[137] A.P. Souri, E. Skliri, I. Vamvasakis, G.S. Armatas & V. Binas. Highly Active Pt Nanoparticles Supported on SrTiO3 for Photocatalytic Hydrogen Production. Applied Physics A, 130, 785 (2024).
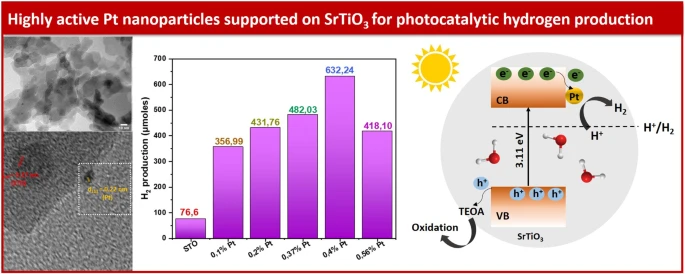
Loading of cocatalysts, and especially noble metal nanoparticles, on the surface of photocatalysts has been proven to be an effective strategy to enhance H2 production, by facilitating charge separation and migration and acting as catalytically active sites. In this work, highly active Pt nanoparticles were loaded with an easy and quick photochemical way on SrTiO3 (STO) and the photocatalytic H2 production was tested under solar and visible irradiation. Characterization techniques such as X-ray diffraction (XRD), scanning electron microscopy, energy dispersive X-ray spectroscopy (SEM/EDX) and transmission electron microscopy (TEM) were used to determine the physical properties of the samples. Additionally, a combination of UV-vis spectroscopy, photoluminescence spectroscopy (PL) and electrochemical impedance spectroscopy (EIS) was used to gain insights into the photocatalytic mechanism and the variation in photocatalytic activity among the samples. Notably, the photocatalyst with the optimum Pt loading (i.e., sample STO-0.4Pt with 0.4 wt% Pt) reached a hydrogen evolution rate of 15,805 µmol g− 1 h− 1, which was found to be ~ 8 times higher than the pristine STO sample under solar irradiation.
[136] E.K. Andreou, I. Vamvasakis, A. Douloumis, G. Kopidakis & G.S. Armatas. Size Dependent Photocatalytic Activity of Mesoporous ZnIn2S4 Nanocrystal Networks. ACS Catal., 14, 14251–14262 (2024).
Understanding of the band-edge electronic structure and charge-transfer dynamics in size-confined nanostructures is vital in designing new materials for energy conversion applications, including green hydrogen production, decomposition of organic pollutants and solar cells. In this study, a series of mesoporous materials comprising continuous networks of linked zinc indium sulfide (ZnIn2S4) nanocrystals with a tunable diameter (ranging from 4 to 12 nm) is reported. These nanomaterials demonstrate intriguing size-dependent electronic properties, charge-transfer kinetics and photocatalytic behaviors. Our extensive characterizations uncover strong size effects on the catalytic activity of constituent ZnIn2S4 nanocrystals in the photochemical hydrogen evolution reaction. As an outcome, the optimized single-component ZnIn2S4 mesostructure produces hydrogen at a 7.8 mmol gcat–1 h–1 release rate under UV-visible light irradiation associated with an apparent quantum yield (AQY) of 17.2% at 420 ±10 nm, far surpassing its microstructured counterpart by a factor of 10.7x. These findings provide a valuable perspective for the rational design of semiconductor nanostructures through synthetic engineering, aiming at the development of high-performance catalysts for zero-carbon energy-related applications.
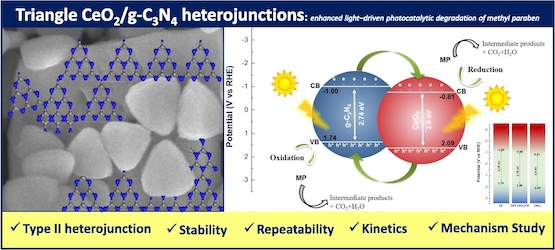
[135] M. Zografaki, S. Stefa, I. Vamvasakis, G.S. Armatas, A.G. Chaidali, I.N. Lykakis & V. Binas. Triangle CeO2/g-C3N4 Heterojunctions: Enhanced Light-Driven Photocatalytic Degradation of Methylparaben. J. Photochem. Photobiol. A, 458, 115976 (2025).
In this work, an efficient photocatalytic system for methylparaben (MP) removal, using solar (λ > 360 nm) and visible (λ > 420 nm) light-driven CeO2/g-C3N4 (CeO2/CN) heterojunctions is reported for the first time. The physicochemical properties of pure CeO2, CN, and CeO2/CN composites were investigated using characterization techniques, such as XRD, FESEM-EDS, TEM, UV–Vis, PL, XPS, and electrochemical spectroscopy. Among the catalysts with different mass ratios of CeO2, 10 %CeO2/CN showed the best photocatalytic performance. This is attributed to the enhanced charge carrier’s separation because of the proper band-edge alignment between CN and CeO2 components, and the strong visible light absorbance. The photocatalytic degradation of MP followed the first-order kinetics, and the 10 %CeO2/CN catalyst exhibited a 3.8- and 11.3-times higher reaction rate (k) constant than that of pure CN, investigated under solar and visible light illumination, respectively. Further, scavenger trapping experiments confirmed that hydroxyl radicals (OH.) and dissolved oxygen are the predominant active species in MP oxidation over 10 %CeO2/CN composite catalyst. 1H NMR and LCMS-HPLC results and observations showed complete degradation of MP (0.1 g/L) to CO2 and H2O after 7 h of solar irradiation, due to the absence of the representative peaks of MP and its organic degradation products (e.g. phenols, benzoates).
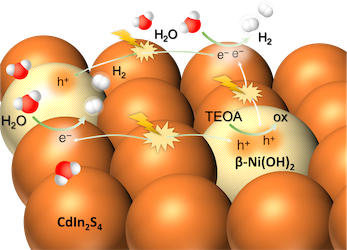
[134] E.K. Andreou, I. Vamvasakis & G.S. Armatas. Fabrication of High-Surface-Area Mesoporous Frameworks of β-Ni(OH)2-CdIn2S4 p-n Nano-heterojunctions for Improved Visible Light Photocatalytic Hydrogen Production. Inorg. Chem. Front., 11, 5273–5285 (2024).
Photocatalytic water splitting holds promise as a cost-effective method for renewable hydrogen production. To this end, synthesising high-performing and robust semiconductor photocatalysts is highly desired. In this study, an interfacial engineering strategy of mesoporous p-n heterojunction frameworks comprising CdIn2S4 assembled nanocrystals (ca. 5–7 nm in size) and β-Ni(OH)2 nanoparticles (ca. 7–8 nm in size) with significantly improved photocatalytic hydrogen evolution activity is described. The promotional effect of β-Ni(OH)2 on the electronic band structure and interfacial charge transfer kinetics of heterostructures is systematically elucidated through a combination of spectroscopic and (photo)electrochemical studies. Incorporating β-Ni(OH)2 effectively accelerates the charge separation process and enhances the utilization of surface-reaching photoexcited carriers for redox reactions. Benefiting from the superior charge transfer and mass transport kinetics, the Ni-modified CdIn2S4 mesostructures evolve H2 at a rate of ~20 mmol h–1 gcat–1 under visible light illumination, corresponding to a 52% apparent quantum efficiency at 420 nm.
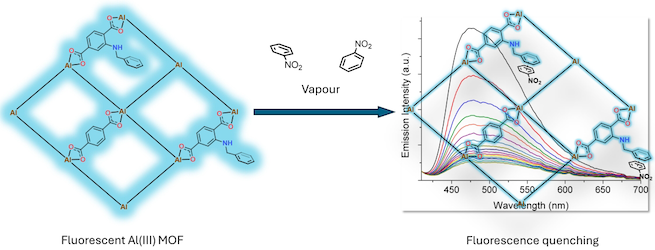
[133] A. Haj-Yahya, D. Kouskouki, A.G. Margellou, E.K. Andreou, G.S. Armatas & T. Lazarides. Functionalised Al(III) Metal Organic Frameworks for Fluorescence Sensing of Nitroaromatic Vapours. J. Mater. Chem. C, 12, 8014–8023 (2024).
The employment of fluorescence sensors provides a platform for rapid and efficient in-field detection of nitroaromatic compounds and is gaining increasing research ground. Herein, we report the synthesis and characterisation of three new fluorescent Al(ΙΙΙ) MOFs, structurally analogous to MIL–53, with the assigned formula {Al(OH)(bdc)1-n(L–1)n}·xsolv (bdc2- = terephthalate; L–1 = 2–((benzyl)amino)-terephthalate). L–1 is a strongly fluorescent dicarboxylic ligand with a pendant π–electron rich aromatic group suitable for donor-acceptor interactions with electron–deficient nitroaromatic guests. Our MOFs show strong fluorescence quenching upon exposure to vapours of nitrobenzene, 1,3-dinitrobenzene, 4-nitrotoluene, 2,4-dinitrotoluene. Additionally, we prepare and study MOF-polymer composites in the form of thin films that are strongly quenched in the presence of nitrobenzene vapours.
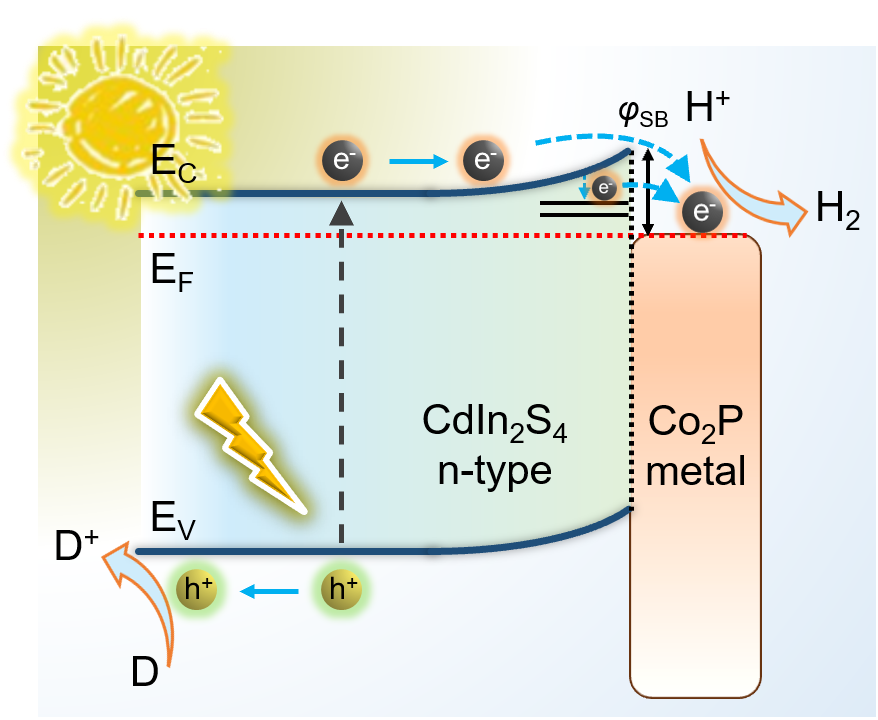
[132] E.K. Andreou, I. Vamvasakis & G.S. Armatas. Efficient Visible-Light Photocatalytic Hydrogen Evolution by Boosting the Interfacial Electron-Transfer in Mesoporous Mott-Schottky Heterojunctions of Co₂P-modified CdIn₂S₄ Nanocrystals. ACS Appl. Energy Mater., 7, 4891–4903 (2024).
Photocatalytic water splitting for hydrogen generation is an appealing means of sustainable solar energy storage. In the last years, mesoporous semiconductors are at the forefront of investigations in low-cost chemical fuel production and energy conversion technologies. Mesoporosity combined with the tunable electronic properties of semiconducting nanocrystals offers the desired large accessible surface and electronic connectivity throughout the framework, thus enhancing photocatalytic activity. In this work, we present the construction of rationally designed 3D mesoporous networks of Co₂P-modified CdIn₂S₄ nanoscale crystals (ca. 5–6 nm in size) through an effective soft-templating synthetic route and demonstrate their impressive performance for visible light-irradiated catalytic hydrogen production. Spectroscopic characterizations combined with electrochemical studies unravel the multi-pathway electron-transfer dynamics across the interface of Co₂P/CdIn₂S₄ Mott-Schottky nano-heterojunctions and shed light on their impact on the photocatalytic hydrogen evolution chemistry. The strong Mott-Schottky interaction occurring at the heterointerface can regulate the charge transport toward greatly improved hydrogen evolution performance. The hybrid catalyst with 10 weight % Co₂P content unveils a H₂ evolution rate of 20.9 mmol gcat⁻¹ h⁻¹ under visible light irradiation with an apparent quantum efficiency (AQE) up to 56.1% at 420 nm, which is among the highest reported activities. The understanding of interfacial charge-transfer mechanism could provide valuable insights into the rational development of highly efficient catalysts for clean energy applications.
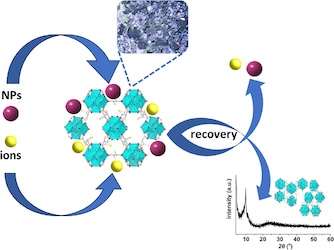
[131] C. Tziasiou, E.K. Andreou, G.S. Armatas, M.J. Manos, A.D. Pournara & D.L. Giokas. Zr⁴⁺-Mercaptosuccinate MOF for the Uptake and Recovery of Gold Nanoparticles and Gold Ions under Batch and Continuous Flow Conditions. Chem. Eng. J, 489, 151107 (2024).
The increasing use of nanomaterials in commercial products has raised concerns regarding their potential effects on water quality and living organisms. So far, most sorbents available for removing nanosized inorganic pollutants from water rely on electrostatic interactions or entrapment in the sorbent pores. However, this limits their applicability in real wastewater samples containing nanomaterials with variable surface properties and sizes, along with high concentrations of competitive species such as inorganic salts and organics. Little attention has also been paid to the recovery of nanoparticles after sorption. In this work, a Zr⁴⁺-mercaptosuccinate metal organic framework (MOF) with free thiol groups was investigated as a sorbent for the removal of Au nanoparticles and Au³⁺ ions from water. Sorption occurs on the surface of the MOF via the formation of strong metal-thiolate chemical bonds enabling the fast uptake of noble metal nanoparticles and noble metal ions from water (within less 1 h). The maximum sorption capacity was found to depend on the size of the Au nanoparticles and ranged from 8 - 41.5 mg/g. The surface functionalization of nanoparticles did not influence sorption performance, which was also maintained in natural waters of variable matrix complexity. The material was also efficient in fixed bed columns with an estimated maximum Au sorption capacity of approximately 7 mg/g, which is significantly higher than the environmental concentrations of Au nanoparticles and adequate for their removal from industrial wastewater. Importantly, the sorbed nanoparticles could be quantitatively recovered (more than 90 %), at the expense of material degradation, enabling their potential reuse.
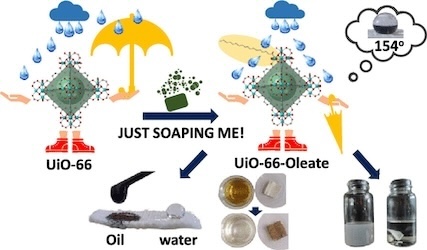
[130] D.A. Evangelou, A.D. Pournara, V.I. Karagianni, C. Dimitriou, E.K. Andreou, Y. Deligiannakis, G.S. Armatas & M.J. Manos. Just Soaping Them: The Simplest Method for Converting Metal Organic Frameworks into Superhydrophobic Materials. ACS Appl. Mater. Interfaces, 16, 12672–12685 (2024).
The incorporation of superhydrophobic properties into metal organic framework (MOF) materials is highly desirable to enhance their hydrolytic stability, gas capture selectivity in the presence of humidity and efficiency in oil–water separations, among others. The existing strategies for inducing superhydrophobicity into MOFs have several weaknesses, such as increased cost, utilization of toxic reagents and solvents, applicability for limited MOFs, etc. Here, we report the simplest, most eco-friendly, and cost-effective process to impart superhydrophobicity to MOFs, involving a rapid (90 min) treatment of MOF materials with solutions of sodium oleate, a main component of soap. The method can be applied to both hydrolytically stable and unstable MOFs, with the porosity of modified MOFs approaching, in most cases, that of the pristine materials. Interestingly, this approach was used to isolate superhydrophobic magnetic MOF composites, and one of these materials formed stable liquid marbles, whose motion could be easily guided using an external magnetic field. We also successfully fabricated superhydrophobic MOF-coated cotton fabric and fiber composites. These composites exhibited exceptional oil sorption properties achieving rapid removal of floating crude oil from water, as well as efficient purification of oil-in-water emulsions. They are also regenerable and reusable for multiple sorption processes. Overall, the results described here pave the way for an unprecedented expansion of the family of MOF-based superhydrophobic materials, as virtually any MOF could be converted into a superhydrophobic compound by applying the new synthetic approach.
[129] E.K. Andreou, I. Vamvasakis & G.S. Armatas. High-Performance Mesoporous Catalysts of Ultrasmall Hexagonal Thiospinel Nanocrystals for Visible-Light Hydrogen Evolution. Adv. Mater. Interfaces, 11, 2300994 (2024).
Semiconductor nanocrystals are at the frontier of energy conversion research owing to their tunable optoelectronic attributes and versatile surface activities. Here we present high-surface-area mesoporous frameworks comprising linked CdIn₂S₄ nanocrystals as efficient catalysts for visible-light-driven hydrogen production. X-ray total scattering analysis discloses hexagonally-structured CdIn₂S₄ thiospinel nanoparticles forming the porous structure. Further analytic results indicate that these newly-made ensembles possess an open-up architecture that is highly conductive and susceptible to modification. Through appropriate selection of the synthesis conditions, we demonstrate that the present synthetic protocol is general, allowing the preparation of porous materials from thiospinel nanoparticles with various sizes and compositions. We show that coupling of CdIn₂S₄ mesostructures and Ni₂P nanosheets substantially expedites the kinetics of water photo-splitting by effectively reinforcing the separation of photogenerated carriers at the interfaces. Thus, mesoporous Ni₂P/CdIn₂S₄ heterojunctions instigate a remarkable improvement in the hydrogen generation rate (~29.3 mmol h⁻¹ gcat⁻¹), presenting an apparent quantum yield of 61.7% at 420 nm monochromatic light. A combination of electrochemical and spectroscopic studies unveils a pertinent mechanistic link between the charge-transfer dynamics and intrinsic photochemical activity in these nanostructures.
(*This article also appears in Hot Topic: Photocatalysis)
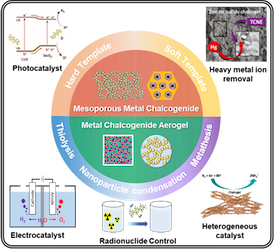
[128] T.D.C. Ha, H. Lee, I. Vamvasakis, G.S. Armatas, Y. Oh & M.-G. Kim. Recent Developments in Porous Metal Chalcogenides for Environmental Remediation and Sustainable Energy. EcoMat, 5, e12419 (2023).
Porous metal chalcogenides have emerged as promising materials for environmental remediation and sustainable energy generation. Their tunable optical band gap (from infrared to the visible range), highly polarizable surface, chemical activity, and adjustable structure make them attractive for various applications. This review summarizes the recent developments concerning the synthesis and characterization of multifunctional porous chalcogenide materials. It explores their remarkable potential in addressing environmental and energy challenges. Moreover, we discuss the several factors that affect the performance of porous metal chalcogenides, such as their microstructure, morphology, and chemical composition, to gain deeper insights into these materials. Finally, we highlight some of the key challenges and future research directions in the development of porous metal chalcogenides as effective and efficient materials for environmental remediation and sustainable energy generation.
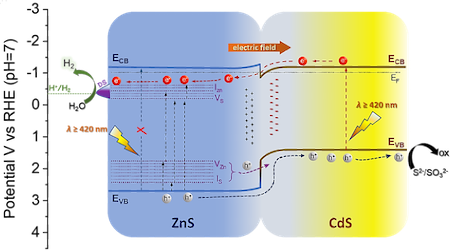
[127] I. Vamvasakis, E.K. Andreou & G.S. Armatas. Mesoporous Dual-Semiconductor ZnS/CdS Nanocomposites as Efficient Visible Light Photocatalysts for Hydrogen Generation. Nanomaterials, 13, 2426 (2023).
The development of functional catalysts for the photogeneration of hydrogen (H₂) through water splitting is crucial in the pursuit of sustainable energy solutions. To that end, metal-sulfide semiconductors, like CdS and ZnS, can play a significant role due to their interesting optoelectronic and catalytic properties. However, the inefficient charge-carrier dissociation and poor photochemical stability are remaining significant limitations to the photocatalytic efficiency. Herein, dual-semiconductor nanocomposites of ZnS/CdS nanocrystal assemblies (NCAs) are developed as efficient visible-light photocatalysts for H₂ generation. The resultant materials, synthesized via a polymer-templated self-polymerization method, comprise a unique combination of ~5-7 nm-sized metal-sulfide nanoparticles that are interlinked to form a 3D open-pore structure with large internal surface area (up to 285 m² g⁻¹) and uniform pores (ca. 6-7 nm). By adjusting the ratio of constituent nanoparticles, the optimized ZnS/CdS catalyst with 50 wt.% ZnS content demonstrates a remarkable stability and visible-light H₂-evolution activity (~29 mmol g⁻¹ h⁻¹ mass activity) with an apparent quantum yield (AQY) of 60% at 420 nm. Photocatalytic evaluation experiments combined with electrochemical and spectroscopic studies suggest that the superior photocatalytic performance of these materials stems from the accessible 3D open-pore structure and the efficient defect-mediated charge transfer mechanism at the ZnS/CdS nanointerfaces. Overall, this work provides a new perspective for designing functional and stable photocatalytic materials for sustainable H₂ production.
(*This article belongs to the Special Issue Early-Career Researchers in Environmental Nanoscience and Nanotechnology)
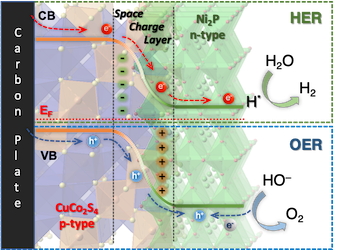
[126] S. Bra, I. Vamvasakis, E.K. Andreou, G. Vailakis, G. Kopidakis & G.S. Armatas. Modulating Interfacial Charge Redistribution of Ni₂P/CuCo₂S₄ p-n Nano-heterojunctions for Efficient Electrocatalytic Overall Water Splitting. Int. J. Hydrogen Energy, 48, 25300–25314 (2023).
Fabricating effective yet inexpensive catalysts is an important target in the research of water electrolysis and clean energy generation. Key challenges still remaining in this area are the rich density of surface-active sites, efficient interfacial charge transfer and improved reaction kinetics. Herein, Ni₂P/CuCo₂S₄ p-n junctions are constructed via an in situ hydrothermal growth of Ni₂P nanoparticles on CuCo₂S₄ nanosheets. Extensive X-ray photoelectron, optical absorption and electrochemical spectroscopy studies coupled with density functional theory calculations provide a mechanistic understanding of the electrochemical behaviour of these catalysts. The integrated Ni₂P/CuCo₂S₄ p-n junctions, owing to the intimate interfacial interactions, offer interesting possibilities to purposively modulate the electronic structure of active sites at the interface, and thus to improve the hydrogen adsorption energetics and electrochemical reaction kinetics. As a result, the catalyst with 30 wt.% Ni₂P content displays high intrinsic electrocatalytic activity, requiring overpotentials of 183 and 360 mV to deliver 10 mA·cm⁻² for HER and 40 mA·cm⁻² for OER in alkaline media, respectively, far lower than those of individual Ni₂P (400 and 520 mV) and CuCo₂S₄ (348 and 380 mV), further showing remarkable durability for 30 h. In addition, an alkaline two-electrode water electrolyzer assembled by Ni₂P/CuCo₂S₄ nano-heterojunctions exhibits a relatively low cell potential of 1.67 V at 10 mA·cm⁻². These Ni₂P-modified CuCo₂S₄ heterostructures demonstrate great potential for renewable hydrogen production technologies, including water electrolysis.
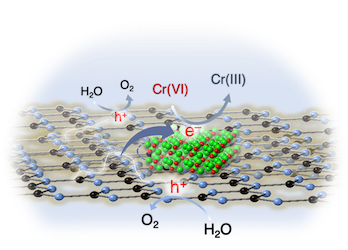
[125] E.K. Andreou, E.D. Koutsouroubi, I. Vamvasakis & G.S. Armatas. Ni₂P-modified P-doped Graphitic Carbon Nitride Hetero-nanostructures for Efficient Photocatalytic Aqueous Cr(VI) Reduction. Catalysts, 13, 437 (2023).
Targeting heterostructures with modulated electronic structures and efficient charge carrier separation and mobility is an effective strategy to improve photocatalytic performance. In this study, we report the synthesis of 2D/3D hybrid heterostructures comprising P-doped graphitic carbon nitride (g-C₃N₄) nanosheets (ca. 50–60 nm in lateral size) and small-sized Ni₂P nanoparticles (ca. 10–12 nm in diameter) and demonstrate their prominent activity in the photocatalytic reduction of Cr(VI). Utilizing a combination of spectroscopic and electrochemical characterization techniques, we unveil the reasons behind the distinct photochemical performance of these materials. We show that Ni₂P modification and P doping of the g-C₃N₄ effectively improve the charge-carrier transportation and spatial separation through the interface of Ni₂P/P-doped g-C₃N₄ junctions. As a result, the catalyst containing 15 wt. % Ni₂P exhibits superior photocatalytic activity in the detoxification of Cr(VI)-contaminated effluents under UV-visible light illumination, presenting an apparent quantum yield (QY) of 12.5% at 410 nm, notably without the use of sacrificial additives. This study marks a forward step in understanding and fabricating cost-effective photocatalysts for photochemical applications.
(*This article belongs to the Special Issue Feature Papers in Environmental Catalysis and selected as one of the Editor's Choice Articles by the MDPI Academic Editors)
[124] P. Georgianos, A.D. Pournara, E.K. Andreou, G.S. Armatas & M.J. Manos. Composite Materials Based on a Zr⁴⁺ MOF and Aluminosilicates for the Simultaneous Removal of Cationic and Anionic Dyes from Aqueous Media. Molecules, 28, 815 (2023).
Environmental pollution has been a reality for many decades, with its contamination intensifying daily due to rapid urbanization and the ever-increasing world population. Dyes, and especially synthetic ones, constitute a category of pollutants that not only affect the quality of water but also exhibit high toxicity toward living organisms. This study was thoroughly planned to explore the removal of two toxic dyes, namely the methylene blue (MB) and methyl orange (MO) compounds from contaminated aqueous media. For this purpose, we designed and synthesized two new composite materials based on ammonium-functionalized Zr⁴⁺ MOF (MOR-1 or UiO-66-NH₃⁺) and naturally occurring sorbents, such as bentonite and clinoptilolite. The composite materials displayed exceptional sorption capability toward both MB⁺ and MO⁻ ions. A key finding of this study was the high efficiency of the composite materials to simultaneously remove MB⁺ and MO⁻ under continuous flow conditions, also showing regeneration capability and reusability, thus providing an alternative to well-known mixed bed resins.
[123] A. Pournara, D. Evangelou, C. Roukounaki, E.K. Andreou, G.S. Armatas, T. Lazarides & M.J. Manos. Highly Efficient Sorption and Luminescence Sensing of Oxoanionic Species by 8-Connected Alkyl-amino Functionalized Zr⁴⁺ MOFs. Dalton Trans., 51, 17301–17309 (2022).
In the present study we provide the sorption properties of four 8-connected Zr⁴⁺ MOFs with the general formula H₁₆[Zr₆O₁₆(RNH-BDC)₄]∙solvent (RNH-BDC²⁻=2-alkyl-amine-terephthalate; R=ethyl-, ET-MOF; R=propyl-, PROP-MOF; R=isobutyl-, SBUT-MOF; R=n-butyl, BUT-MOF) towards toxic Cr(VI) and radionuclide-related ReO₄⁻ oxoanions. These MOFs represent superior sorbents for the removal of the oxoanionic species, in terms of kinetics, sorption isotherms, selectivity and regeneration/reusability. The excellent sorption capability of the MOFs is due to a combination of surface and intraframework sorption phenomena. The latter process proceeds via replacement of terminal water/hydroxyl ligands from the Zr₆ clusters and subsequent binding of oxonanions to the Zr⁴⁺ centers, a fact that was proved via Rietveld PXRD analysis for the anion-loaded BUT-MOF. Importantly, BUT-MOF demonstrated an expectional sorption capacity for Cr₂O₇²⁻ (505 mg/g) and was further utilized in a sorption column in the form of MOF/calcium alginate beads, displaying remarkable removal efficiency towards industrial (chrome-plating) wastewater. Furthermore, the luminescence Cr(VI) sensing properties of BUT-MOF were explored in detail, presenting high sensitivity (detection limits as low as 9 ppb) and selectivity for these species against various competitive anions.
[122] A. Pournara, S. Rizogianni, D. Evangelou, E.K. Andreou, G.S. Armatas & M.J. Manos. Zr⁴⁺-Terephthalate MOFs with 6-Connected Structures, Highly Efficient As(III/V) Sorption and Superhydrophobic Properties. Chem. Commun., 58, 8862–8865 (2022).
The use of terephthalate ligands with C𝑛H2𝑛+1NH-chains (𝑛≥6) led to the isolation of the first examples of Zr4+-terephthalate MOFs with 6-connected frameworks. The material with hexyl-amino functional groups has been proved to be an exceptional sorbent for the removal of As(III/V) toxic species from aqueous media, whereas MOFs with heptyl to dodecyl-amino moieties are superhydrophobic with promising oil-water separation properties.
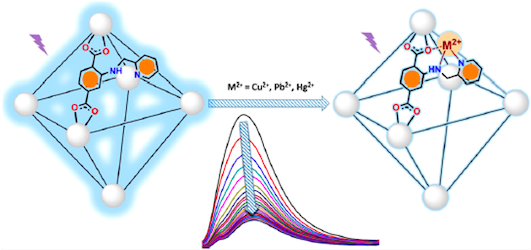
[121] S.A. Diamantis, A.D. Pournara, E.D. Koutsouroubi, C. Moularas, Y. Deligiannakis, G.S. Armatas, A.G. Hatzidimitriou, M.J. Manos & Th. Lazarides. Detection and Sorption of Heavy Metal Ions in Aqueous Media by a Fluorescent Zr(IV) Metal−Organic Framework Functionalized with 2-Picolylamine Receptor Groups. Inorg. Chem., 61, 7847–7858 (2022).
Increasing global environmental pollution due to heavy metal ions raises the importance of research on new multifunctional materials for simultaneous detection and removal of these contaminants from water resources. In this study, we report a microporous 8-connected Zr⁴⁺ metal−organic framework (MOF) based on a terephthalate ligand decorated with a chelating 2-picolylamine side group (dMOR-2), which shows highly efficient fluorescence sensing and sorption of heavy metal cations. We demonstrate by detailed fluorescence studies the ability of a water-dispersible composite of dMOR-2 with polyvinylpyrrolidone for real-time detection of Cu²⁺, Pb²⁺, and Hg²⁺ in aqueous media. The limits of detection were found to be below 2 ppb for these species, while the system’s performance is not affected by the presence of other potentially competitive ions. In addition, sorption studies showed that a composite of dMOR-2 with calcium alginate (dMOR-2@CaA) is an excellent sorbent for Pb²⁺ and Cu²⁺ ions with capacities of 376 ± 15 and 117 ± 4 mg per gram of dMOR-2@CaA, respectively, while displaying the capability for simultaneous removal of various heavy metal ions in low initial concentrations and in the presence of large excesses of other cationic species. Structural and spectroscopic studies with model ligands analogous to our material’s receptor unit showed chelation to the 2-picolylamine moiety to be the main binding mode of metal ions to dMOR-2. Overall, dMOR-2 is shown to represent a rare example of a MOF, which combines sensitive fluorescence detection and high sorption capacity for heavy metal ions.
[120] A.D. Pournara, E.K. Andreou, G.S. Armatas & M.J. Manos. Zirconium(IV) Metal Organic Frameworks with Highly Selective Sorption for Diclofenac under Batch and Continuous Flow Conditions. Crystals, 12, 424 (2022).
Diclofenac (DCF) is one of the most effective non-steroidal anti-inflammatory drugs (NSAIDs) and at the same time one of the most consumed drugs worldwide. Since the ever-increasing use of diclofenac poses serious threats to ecosystems, its substantial removal is crucial. To address this issue a variety of sorbents has been employed. Herein we report the diclofenac sorption properties of two metal organic frameworks, namely [Zr₆O₄(OH)₄(NH₂BDC)₆]·xH₂O (MOR-1) and H₁₆[Zr₆O₁₆(H₂PATP)₄]·xH₂O (MOR-2). Batch studies revealed fast sorption kinetics for removal of DCF⁻ from water as well as particularly high selectivity for the drug vs. common competitive species. Furthermore, MOR-1, in its composite form with alginic acid, was utilized in a sorption column, displaying remarkable removal efficiency towards DCF⁻ anions. Significantly, this is the first time that column sorption data for removal of NSAIDs using MOF-based materials is reported.
(*This article belongs to the Special Issue Organic-Inorganic Hybrids: Synthesis, Property and Application)
[119] D.A. Evangelou, A.D. Pournara, Ch. Tziasiou, E.K. Andreou, G.S. Armatas & M.J. Manos. A Robust Al³⁺ MOF with Selective As(V) Sorption and Efficient Luminescence Sensing Properties Towards Cr(VI). Inorg. Chem., 61, 2017–2030 (2022).
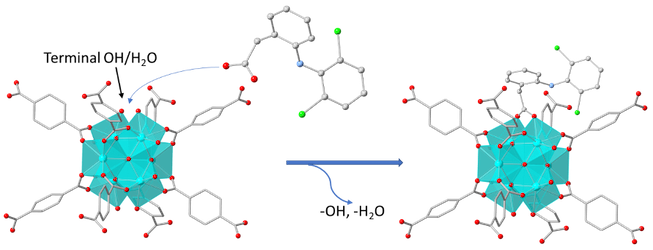
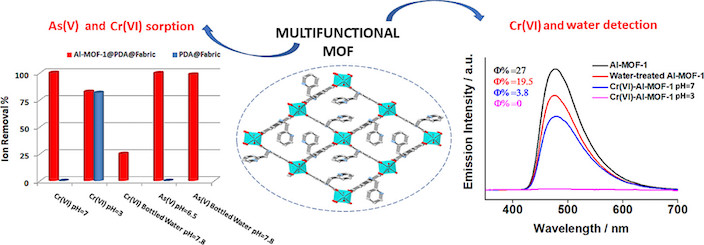
Herein, we report the synthesis and characterization of a new robust Al³⁺ MOF, [Al(OH)(PATP)]·solvent (Al-MOF-1, with PATP²⁻ = 2-((pyridin-2-ylmethyl)amino)terephthalate). Al-MOF-1 was proved to be an outstanding sorbent towards As(V) with excellent sorption capacity (71.9 ± 3.8 mg As/g), extraordinary sorption kinetics (equilibrium time ≤ 1 min) and ultra-high selectivity in the presence of various competing anions. Furthermore, Al-MOF-1 revealed high sorption capacities for Cr(VI) species in both neutral (124.5 ± 8.6 mg Cr/g) and acidic (63 ± 2 mg Cr/g) aqueous media, combining fast kinetics and relatively good selectivity. Aiming in practical wastewater treatment, we have also immobilized Al-MOF-1 on a cotton substrate, coated with polydopamine. The fabric sorbent exhibited highly effective removal of the toxic oxoanionic species from aqueous media under either batch or dynamic (continuous flow) conditions. In addition, Al-MOF-1 was found to be promising sensor for detecting trace amounts of Cr(VI) in real water samples, characterized by high sensitivity and selectivity, with Cr(VI) being successfully detected at concentrations well below the acceptable limits (< 50 ppb). Moreover, Al-MOF-1 was found to be a sufficient water sensor in organic solvents (LOD ≤ 0.25% v/v). All the above indicate that Al-MOF-1 represents a multifunctional material with a multitude of potential applications, such as environmental remediation, industrial wastewater treatment, chemical analysis and water determination in biofuels.
[118] Ch. Patriarchea, I. Vamvasakis, E.D. Koutsouroubi & G.S. Armatas. Enhancing Interfacial Charge Transfer in Mesoporous MoS₂/CdS Nanojunction Architectures for Highly Efficient Visible-Light Photocatalytic Water Splitting. Inorg. Chem. Front., 9, 625–636 (2022).
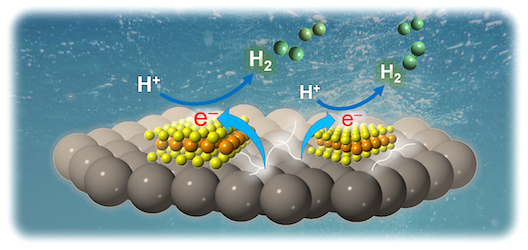
Modulating the heterojunction interfaces at the nanoscale is paramount to developing effective photocatalysts. Nanodimensional heterostructures, essentially, expose a large fraction of active edge sites, which, along with excellent electronic communication, can result in high solar energy to chemical fuel conversion efficiencies. In this work, mesoporous heterojunction architectures made of ultrasmall MoS₂ nanolayers (ca. 10–15 nm in lateral size) and sub 5 nm sized CdS nanocrystals have been prepared thought a polymer-templated oxidative aggregation of CdS nanocrystal followed by a wet-chemical deposition of exfoliated MoS₂ nanosheets on CdS surface. Thorough chemical, morphological and structural investigations by electron microprobe analysis, X-ray diffraction, electron microscopy, X-ray photoelectron spectroscopy and N₂ physisorption prove that the MoS₂-modified CdS nanocatalysts are composed of a porous network of connected cubic CdS nanocrystals and 2H-phase MoS₂ nanosheets and possess high internal BET surface area (ca. 159–225 m² g⁻¹) and uniform pores (ca. 6–9 nm in diameter). Photocatalytic studies coupled with UV–vis/NIR, photoluminescence and electrochemical impedance spectroscopy measurements indicate that the nanoscale MoS₂/CdS junctions provide a more efficient electronic connectivity and charge carrier dissociation across the catalyst interfaces, resulting in a remarkable enhancement in the photocatalytic H₂ production activity. The optimized MoS₂/CdS catalyst at 20 wt.% MoS₂ content reaches a H₂ production rate up to ~0.4 mmol h⁻¹ (or ~19 mmol h⁻¹ g⁻¹ mass activity) with remarkable stability under visible light irradiation, corresponding to an overall 6.7x enhancement of H₂ generation efficiency relative to the unmodified CdS. We also obtained an apparent quantum yield (AQY) of 51.2% for hydrogen generation reaction using monochromatic light of 420 nm.
[117] E.D. Koutsouroubi, I. Vamvasakis, Ch. Drivas, S. Kennou & G.S. Armatas. Photochemical Deposition of SnS₂ on Graphitic Carbon Nitride for Photocatalytic Aqueous Cr(VI) Reduction. Chem. Eng. J. Adv., 9, 100224 (2022).
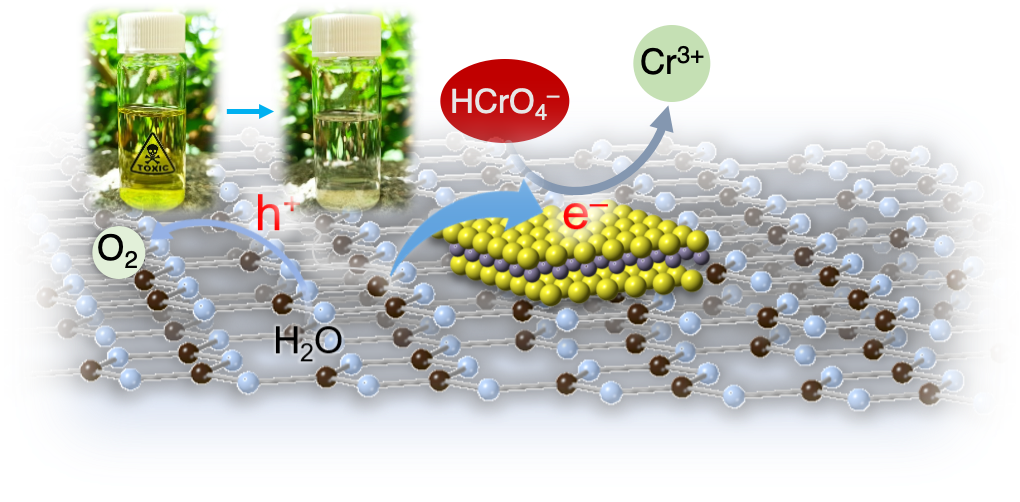
Cost-effective and efficient photocatalysts are in demand for high-performance clean energy and environmental technologies. In this article, we report the synthesis of 2D/2D SnS₂/g-C₃N₄ layered heterostructures with reduced interfacial resistance and improved charge transfer kinetics via a photochemical deposition method. The SnS₂/g-C₃N₄ catalysts demonstrate outstanding photocatalytic Cr(VI) reduction (with a 21.2 μmol h⁻¹ Cr(VI) consumption rate) and water oxidation (with a 15.1 μmol h⁻¹ O₂ evolution rate) activity, reaching energy conversion efficiencies of up to 16.4% and 12.1% at 375 nm and 410 nm, respectively, that is among the best known Cr(VI) reduction catalysts reported to date. Based on X-ray photoelectron and UV–vis/NIR absorption spectroscopy, and electrochemical and photoelectrochemical measurements, we provide a mechanistic understanding of the redox reactions and charge transport dynamics in this catalytic system. The results demonstrate the potential viability of the SnS₂-decorated g-C₃N₄ nano-heterostructures for environmental remediation applications, including decontamination of Cr(VI) polluted aquatic systems.
[116] A. Ioakeimidis, I.T. Papadas, E.D. Koutsouroubi, G.S. Armatas & S.A. Choulis. Thermal Analysis of Metal-Organic Precursors for Functional Cu:NiOx Hole Transporting Layer in Inverted Perovskite Solar Cells: Role of Solution Combustion Chemistry in Cu:NiOx Thin Films Processing. Nanomaterials, 11, 3074 (2021).
Low temperature solution combustion synthesis emerges as a facile method for synthesis of functional metal oxides thin films for electronic applications. We study the solution combustion synthesis process of Cu:NiOx using different molar ratios (w/o, 0.1 and 1.5) of fuel acetylacetone (Acac) to oxidizer (Cu, Ni Nitrates) as a function of thermal annealing temperatures 150, 200 and 300 ᴼC. The solution combustion synthesis process, in both thin films and bulk Cu:NiOx, is investigated. Thermal analysis studies reveal that the Cu:NiOx thin films show a more gradual mass loss while the bulk Cu:NiOx exhibits a distinct combustion process. The thin films can crystallize to Cu:NiOx at annealing temperature of 300 ᴼC irrespective to the Acac concentration whereas lower an-nealing temperatures (150 and 200 ᴼC) produce amorphous materials. Finally, 50 nm Cu:NiOx thin films are introduced as HTLs within the inverted perovskite solar cell device architecture. The Cu:NiOx HTL annealed at 150 and 200 ᴼC provided PVSCs with limited functionality whereas efficient triple-cation Cs₀.₀₄(MA₀.₁₇FA₀.₈₃)₀.₉₆ Pb(I₀.₈₃Br₀.₁₇)₃ based PVSCs achieved for Cu:NiOx HTLs annealed at temperature 300 ᴼC.
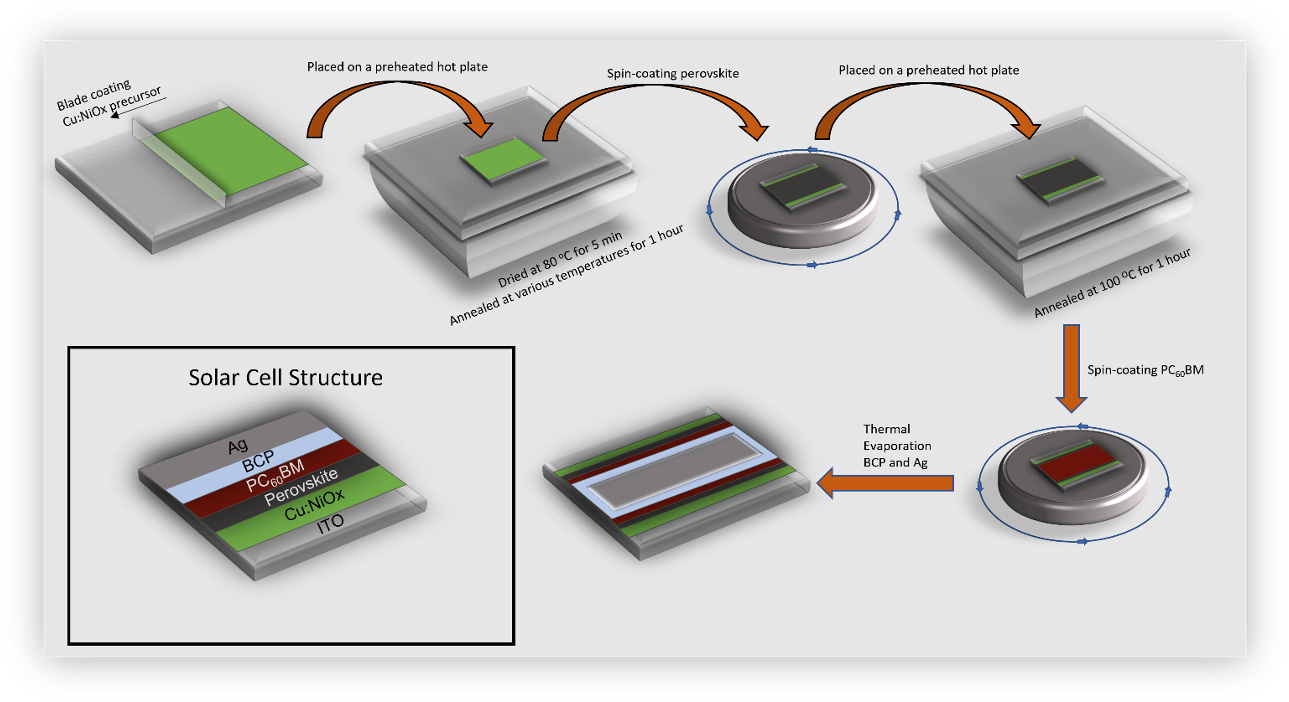
(*This article belongs to the Special Issue Advances in Nanomaterials for Perovskite Solar Cells)
[115] V. Daikopoulou, E. Skliri, E.D. Koutsouroubi, G.S. Armatas & I.N. Lykakis. Selective Mild Oxidation of Anilines into Nitroarenes via Catalytic Activation of Mesoporous Frameworks Linked with Gold-loaded Mn₃O₄ Nanoparticles. ChemPlusChem, 87, e202100413 (2022).
In this article, we report the synthesis and catalytic application of mesoporous Au-loaded Mn₃O₄ nanoparticle assemblies (MNAs) with different Au contents, i.e., 0.2, 0.5 and 1 wt %, towards the selective oxidation of anilines into the corresponding nitroarenes. Among common oxidants, as well as several supported gold nanoparticle platforms, Au/Mn₃O₄ MNAs containing 0.5 wt % Au with an average particle size of 3–4 nm show the best catalytic performance in the presence of tert-butyl hydroperoxide (TBHP) as a mild oxidant. In all cases, the corresponding nitroarenes were isolated in high to excellent yields (85–97%) and selectivity (>98%) from acetonitrile or greener solvents, such as ethyl acetate, after simple flash chromatography purification. The 0.5% Au/Mn₃O₄ catalyst can be isolated and reused several times without a significant loss of its catalytic activity and can be applied successfully to a lab-scale reaction. Based on the observed high chemoselectivity and isolated yields, and the fast and clean reaction processes, the present catalytic system Au/Mn₃O₄–TBHP shows promise for the efficient synthesis of aromatic nitro compounds at a large scale and eco-sustainable level.
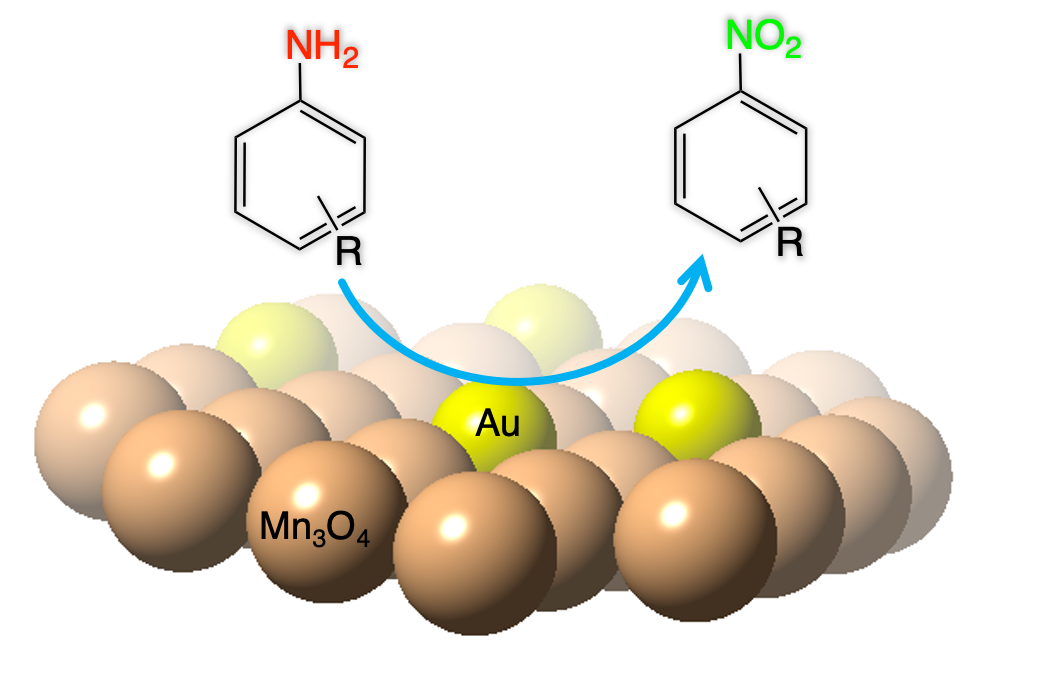
(*This article also appears in special collections: Gold, Chem. Eur. J., 2021 and 10th Anniversary of ChemPlusChem, ChemPlusChem, 87, 2022)
[114] I.T. Papadas, A. Ioakeimidis, I. Vamvasakis, P. Eleftheriou, G.S. Armatas & S.A. Choulis. All-Inorganic p−n Heterojunction Solar Cells by Solution Combustion Synthesis using n-type FeMnO₃ Perovskite within the Photoactive Layer. Front. Chem., 9, 754487 (2021).
This study outlines the synthesis and physicochemical characteristics of a solution-processable iron manganite (FeMnO₃) nanoparticles via a chemical combustion method using tartartic acid as a fuel and demonstrates the performance of this material as a n-type photoactive layer in all-oxide solar cells. It is shown that the solution combustion synthesis (SCS) method enables the formation of pure crystal phase FeMnO₃ with controllable particle size. Moreover, to assemble a network of connected FeMnO₃ nanoparticles, β-alanine was used as a capping agent and dimethylformamide (DMF) as a polar aprotic solvent for the colloidal dispersion of FeMnO₃ NPs. This procedure yields a ~500 nm thick photoactive layer. The proposed method is crucial to obtain functional solution processed NiO/FeMnO₃ heterojunction inorganic photovoltaics. Photovoltaic performance and solar cell device limitations of the NiO/FeMnO₃-based heterojunction solar cells are presented.
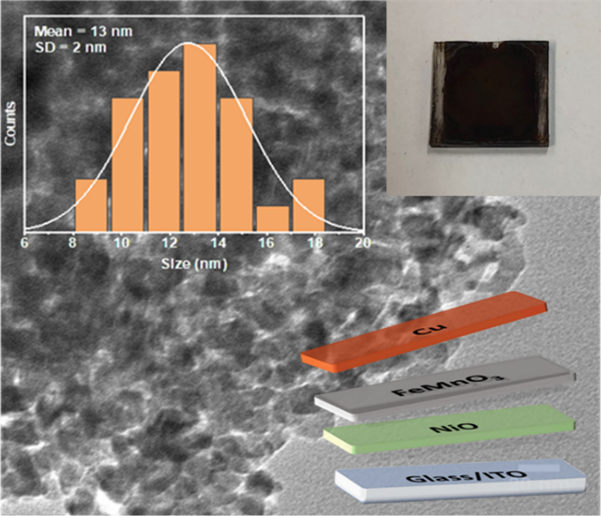
(*This article is part of the research topic: Design, Synthesis and Applications of Novel Photoactive Nanomaterials)
[113] E.D. Koutsouroubi, I. Vamvasakis, M.G. Minotaki, I.T. Papadas, Ch. Drivas, S.A. Choulis, G. Kopidakis, S. Kennou & G.S. Armatas. Ni-doped MoS₂ Modified Graphitic Carbon Nitride Layered Hetero-nanostructures as Highly Efficient Photocatalysts for Environmental Remediation. Appl. Catal. B: Environ., 297, 120419 (2021).
Highly efficient and cost-effective photocatalysts are among the most prominent targets in the field of environmental remediation and clean energy production. Here, we report that 2D/2D layer heterostructures composed of exfoliated Ni-doped MoS₂ nanosheets and g-C₃N₄ layers can carry out photocatalytic Cr(VI) reduction in aqueous solutions with outstanding activity, exhibiting apparent QYs as high as 29.6 % and 23.7 % at 375 and 410 nm. We show that Ni doping of MoS₂ markedly increases the photochemical activity, which, together with electrochemical, spectroscopic and theoretical DFT studies, arises from the enhanced carrier density and mobility at the Ni-MoS₂/g-C₃N₄ interface. In addition to the favorable charge transport properties, delineation of the photoinduced oxidation reactions by gas monitoring techniques reveals that the high efficiency also arises from fast water oxidation kinetics. The results of this work mark an important step forward in understanding and designing low-cost and earth-abundant catalysts for detoxification of Cr(VI)-contaminated industrial effluents.
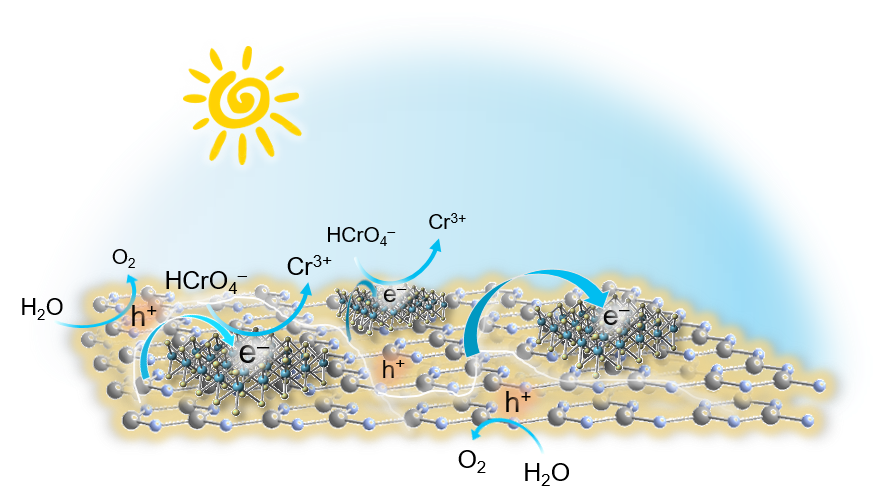
[112] E. Skliri, I. Vamvasakis, I.T. Papadas, S.A. Choulis & G.S. Armatas. Mesoporous Composite Networks of Linked MnFe₂O₄ and ZnFe₂O₄ Nanoparticles as Efficient Photocatalysts for the Reduction of Cr(VI). Catalysts, 11, 199 (2021).
Semiconductor photocatalysis has recently emerged as an effective and eco-friendly approach that could meet the stringent requirements for sustainable environmental remediation. To this end, the fabrication of novel photocatalysts with unique electrochemical properties and high catalytic efficiency is of utmost importance and requires adequate attention. In this work, dual com-ponent mesoporous frameworks of spinel ferrite ZnFe₂O₄ (ZFO) and MnFe₂O₄ (MFO) nanoparticles are reported as efficient photocatalysts for detoxification of hexavalent chromium (Cr(VI)) and organic pollutants. The as-prepared materials, which are synthesized via a polymer-templated aggregating self-assembly method, consist of a continuous network of linked nanoparticles (ca. 6–7 nm) and exhibit large surface area (up to 91 m² g⁻¹) arising from interstitial voids between the nanoparticles, according to electron microscopy and N₂ physisorption measurements. By tuning the composition, MFO-ZFO composite catalyst containing 6 wt.% MFO at-tains excellent photocatalytic Cr(VI) reduction activity in the presence of phenol. In-depth studies with UV-visible absorption, electrochemical and photoelectrochemical measurements show that the performance enhancement of this catalyst predominantly arises from the suitable band edge positions of constituent nanoparticles that efficiently separates and transports the charge carriers through the interface of the ZFO/MFO junctions. Besides, the open pore structure and large surface area of these ensembled networks also boost the reaction kinetics. The remarkable activity and durability of the MFO-ZFO heterostructures implies the great possibility of implementing these new nanocomposite catalysts into a realistic Cr(VI) detoxification of contaminated wastewater.
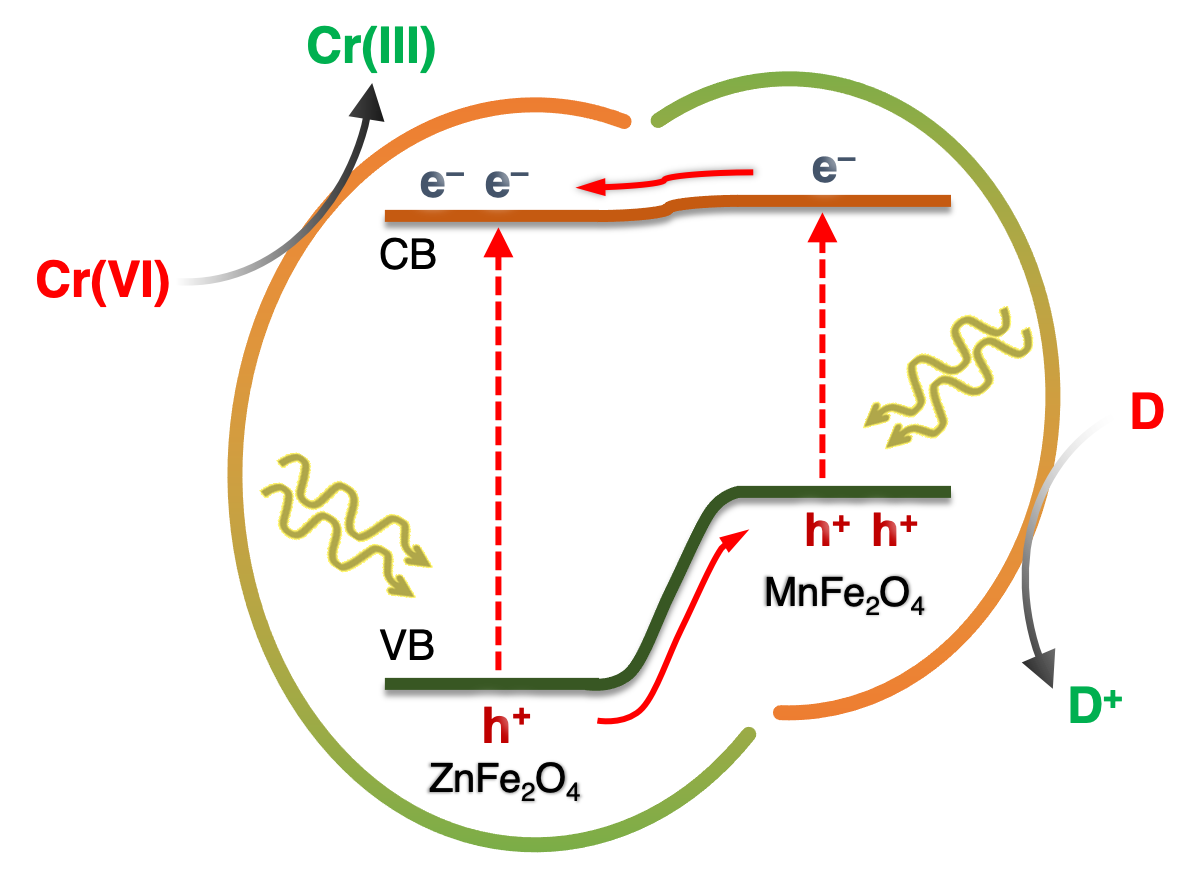
(*This article is part of the special issue: Rational Design of Non-precious Metal Oxide Catalysts by Means of Advanced Synthetic and Promotional Routes)
[111] A. Pournara, S. Rapti, A. Valmas, I. Margiolaki, E.K. Andreou, G.S. Armatas, A. Tsipis, J. Plakatouras, D.L. Giokas & M.J. Manos. Alkylamino-Terephthalate Ligands Stabilize 8-Connected Zr⁴⁺ MOFs with Highly Efficient Sorption for Toxic Se Species. J. Mater. Chem. A, 9, 3379–3387 (2021).
Zirconium(IV) metal organic frameworks (MOFs) with low net connectivity and available intra-framework sorption sites constitute excellent sorbents for toxic anions. To expand this family of highly promising sorbents, it would be desired to develop new synthetic strategies aiming in such materials. Here we show that the utilization of terephthalate ligands with small to medium size alkyl-amino functional groups comprises an effective approach towards microporous Zr⁴⁺ MOFs with 8-connected frameworks. The new MOFs were proved the most effective Se(IV) and Se(VI) sorbents ever reported, with exceptional sorption capacities (up to 272 mg Se(IV)/g and 290 mg Se(VI)/g), reusability, rapid sorption kinetics (≤ 3 min) and capability to sorb efficiently these anions in a very wide pH range (1-10), even in the presence of various competitive anions. The MOFs also display highly efficient sorption capability for the particularly toxic SeCN⁻, with such property first time demonstrated for MOF materials.
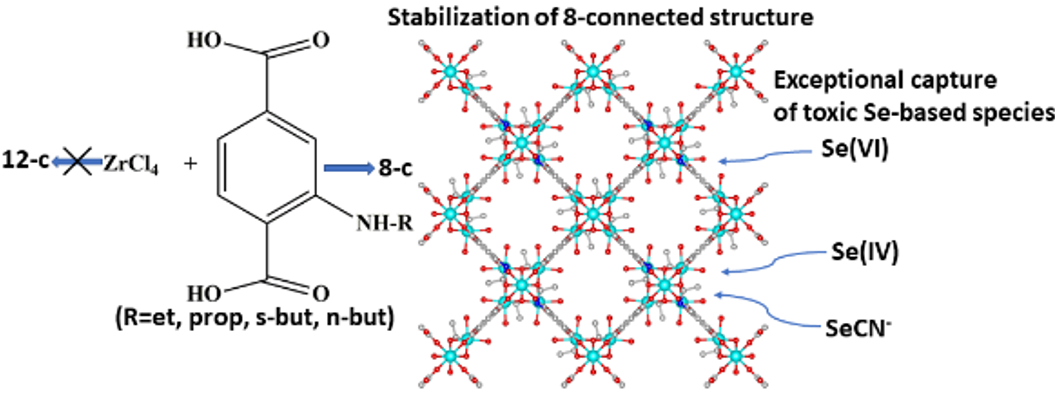
(*This article is part of the themed collection:Journal of Materials Chemistry A HOT Papers)
[110] V. Nikolaou, G. Charalambidis, K. Ladomenou, E. Nikoloudakis, Ch. Drivas, I. Vamvasakis, S. Panagiotakis, G. Landrou, E. Agapaki, C. Stangel, C. Henkel, J. Joseph, G.S. Armatas, M. Vasilopoulou, S. Kennou, D.M. Guldi & A.G. Coutsolelos. Controlling Solar Hydrogen Production by Organizing Porphyrins. ChemSusChem, 14, 961–970 (2021).
In this study, a highly efficient photocatalytic H₂ production system is developed by employing porphyrins as photocatalysts. Palladium and platinum tetracarboxyporphyrins (PdTCP and PtTCP) are adsorbed or coadsorbed onto TiO₂ nanoparticles (NPs), which act as the electron transport medium and as a scaffold that promotes the self‐organization of the porphyrinoids. The self‐organization of PdTCP and PtTCP, forming H‐ and J‐aggregates, respectively, is the key element for H₂ evolution, as in the absence of TiO₂ NPs no catalytic activity is detected. Notably, J‐aggregated PtTCPs are more efficient for H₂ production than H‐aggregated PdTCPs. In this approach, a single porphyrin, which self‐organizes onto TiO₂ NPs, acts as the light harvester and simultaneously as the catalyst, whereas TiO₂ serves as the electron transport medium. Importantly, the concurrent adsorption of PdTCP and PtTCP onto TiO₂ NPs results in the most efficient catalytic system, giving a turnover number of 22,733 and 30.2 mmol(H₂) g(cat)⁻¹.
[109] I. Daskalakis, I. Vamvasakis, I.T. Papadas, S. Tsatsos, S.A. Choulis, S. Kennou & G.S. Armatas. Surface Defect Engineering of Mesoporous Cu/ZnS Nanocrystal-Linked Networks for Improved Visible-Light Photocatalytic Hydrogen Production. Inorg. Chem. Front., 7, 4687–4700 (2020).
Transition metal sulfides have been emerging as one of the most attractive and prospective catalysts for direct conversion of solar energy into chemical fuels. The intriguing compositional and electronic characteristics and their feasibility for integration in porous architectures endow metal sulfide materials with superior activity for photochemical catalysis. In the present work, high-surface-area Cu-doped ZnS nanocrystal (NC)-linked mesoporous frameworks are successfully synthesized for use as cost-effective catalysts for photochemical hydrogen evolution. Benefiting from the proper band-edge alignment and enhanced visible light absorption resulted from interfacial charge transfer between ZnS and Cu₂S NCs, there is a spatial separation of charge carries which leads to excellent activity for the photocatalytic hydrogen production. Moreover, results here show that surface defect passivation through a wet-chemical sulfidation process effectively increases the photochemical performance of the composite catalysts by improving the transport efficiency of electrons at the Cu₂S/ZnS interface and changing the Helmholtz layer potential drop at the ZnS/Cu₂S/electrolyte junction. Thus, a remarkably improvement of 1 mmol h⁻¹ gcat⁻¹ for hydrogen evolution is observed with sulfide-treated Cu₂S/ZnS catalyst containing 5 mol% Cu, which is associated with a 17.6% apparent quantum yield under 410 nm irradiation. This work marks an interesting strategy for enhancing interface charge transfer properties and hydrogen evolution activity of metal sulfides by surface defect engineering with sulfide ions.
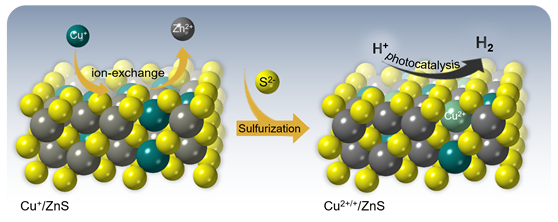
[108] L. Du, D. Feng, X. Xing, C. Wang, G.S. Armatas & D. Yang. Uniform Palladium-Nickel Nanowires Arrays for Stable Hydrogen Leakage Detection and Efficient Hydrogen Evolution Reaction. Chem. Eng. J., 400, 125864 (2020).
As a high-energy-density, renewable and green energy carrier, hydrogen has recently received increasing attention. However, the reliable detection for hydrogen leakage, especially in harsh environmental conditions, and the efficient high-purity hydrogen production are still challenging issues. Here, we report that uniform palladium / nickel nanowires (PdNi NWs) arrays, which have been synthesized via anode-aluminum-oxide (AAO) template-confined electrodeposition as bi-functional material for hydrogen-leakage detection and hydrogen evolution reaction (HER). PdNi NWs arrays exhibit rough surface with the diameter of ~ 60 nm and present alloyed crystallization. As hydrogen sensing materials, PdNi NWs enable to stably work within wide temperature range of 153 - 388 K, for instance 0.1% hydrogen can be detected at 213 K, in which Pd alloyed with Ni effectively decreases the critical temperature of “reverse sensing behavior” to 153 K. Meanwhile, benefited from the synergic effect of exposing more active sites by the rough surface and modifying the electronic structure of Pd by Ni incorporation, PdNi NWs show the overpotential of 91 mV at 10 mA / cm², and the Tafel slope of 96 mV / dec in 0.5 M H₂SO₄. The PdNi NWs developed in this study show great perspectives for the safe detection and high-purity production of hydrogen.
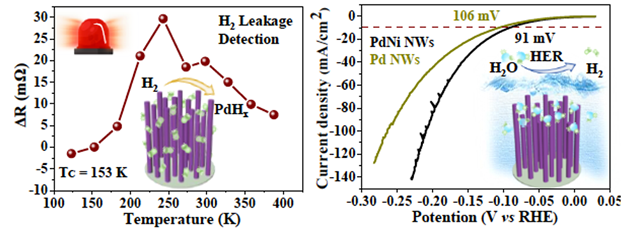
[107] Y. Georgiou, S. Rapti, A. Mavrogiorgou, G.S. Armatas, M.J. Manos, M. Louloudi & Y. Deligiannakis. A Hybrid {Silk@Zirconium MOF} Material as Highly Efficient AsIII-sponge. Sci. Rep., 10, 9358 (2020).
Exposure of humans to Arsenic from groundwater drinking sources is an acute global public health problem, entailing the urgent need for highly efficient/low-cost Arsenite (Asᴵᴵᴵ) up-taking materials. Herein we present an innovative hybrid-material, ZrMOF@SFd operating like an “Asᴵᴵᴵ-sponge” with unprecedented efficiency of 1800 mg Asᴵᴵᴵ gr⁻¹. ZrMOF@SFd consists of a neutral Zirconium Metal-Organic Framework [ZrMOF] covalently grafted on a natural silk-fiber (SFd). ZrMOF itself exhibits Asᴵᴵᴵ adsorption of 2200 mg gr⁻¹, which supersedes any -so far- known Asᴵᴵᴵ-sorbent. Using XPS, FTIR, BET-porosimetry data, together with theoretical Surface-Complexation-Modeling (SCM), we show that the high-Asᴵᴵᴵ-uptake is due to a sequence of two phenomena: [i] at low Asᴵᴵᴵ-concentrations, surface-complexation of H₃AsO₃ results in Asᴵᴵᴵ-coated voids of ZrMOF, [ii] at increased Asᴵᴵᴵ-concentrations, the Asᴵᴵᴵ-coated voids of ZrMOF are filled-up by H₃AsO₃ via a partitioning-like mechanism. In a more general context, the present research exemplifies a mind-changing concept, i.e. that a “partitioning-like” mechanism can be operating for adsorption of metalloids, such as H₃AsO₃, by metal oxide materials. So far, such a mechanism has been conceptualized only for the uptake of non-polar organics by natural organic matter or synthetic polymers.
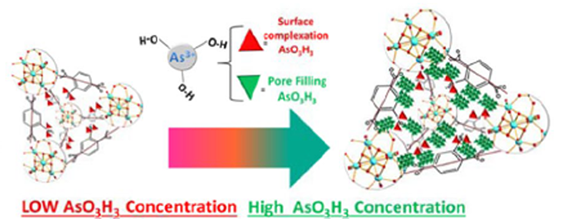
[106] P. Psathas, Y. Georgiou, C. Moularas, G.S. Armatas & Y. Deligiannakis. Controlled-Phase Synthesis of Bi₂Fe₄O₉ & BiFeO₃ by Flame Spray Pyrolysis and their Evaluation as Non-noble Metal Catalysts for Efficient Reduction of 4-Nitrophenol. Powder Technol., 368, 268–277 (2020).
Pure phases of Bismuth-Iron-Oxide nanomaterials, i.e. perovskite BiFeO₃-phase or mullite-type Bi₂Fe₄O₉-phase have been synthesized, using Flame Spray Pyrolysis (FSP). Based on a systematic study of the phase-formation by XRD diffraction, X-Ray Photoelectron and Raman spectroscopies, FSP protocols are developed for the synthesis of 100% pure phases of either BiFeO₃ or Bi₂Fe₄O₉, as well as mixed-phase [BiFeO₃: Bi₂Fe₄O₉] at controllable phase-percentages. The materials are shown to be quite efficient in catalytic reduction of 4-nitrophenol to 4-aminophenol in the presence of NaBH₄. Based on an Arrhenius study, the activation energy for the of 4-nitrophenol catalysis is estimated Ea = 22 kJ mol⁻¹ for BiFeO₃ and Ea = 36 kJ mol⁻¹ for Bi₂Fe₄O₉. Thus, the FSP-made BiFeO₃-nanophase shows enhanced catalytic reaction of 4-nitrophenol that is comparable to the, so far reported, best-performing noble-metal nanocatalysts. The catalytic physicochemical mechanism is discussed based on the thermodynamic, in-situ FT-IR, and 4-nitrophenol adsorption isotherms data for the BiFeO₃, Bi₂Fe₄O₉ nanophases.
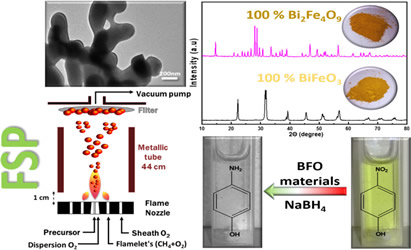
[105] E.D. Koutsouroubi, I. Vamvasakis, I.T. Papadas, Ch. Drivas, S.A. Choulis, S. Kennou & G.S. Armatas. Interface Engineering of MoS₂‐Modified Graphitic Carbon Nitride Nano‐photocatalysts for an Efficient Hydrogen Evolution Reaction. ChemPlusChem, 85, 1379–1388 (2020).
Understanding of photochemical charge transfer processes on the nanoscale heterojunctions is essential in developing effective catalysts. Here, we utilize a controllable synthesis method and a combination of optical absorption, photoluminescence, and electrochemical impedance spectroscopic studies to investigate the effect of MoS₂ nanosheet lateral dimension and edge length size on the photochemical behavior of MoS₂‐modified graphitic carbon nitride (g‐C₃N₄) heterojunctions. These nano‐heterostructures, which comprise interlayer junctions with variable area (i.e., MoS₂ lateral size ranges from 18 nm to 52 nm), provide a size‐tunable interfacial charge transfer through the MoS₂/g‐C₃N₄ contacts, while exposing a large fraction of surface MoS₂ edge sites available for hydrogen evolution reaction. Importantly, modification of g‐C₃N₄ with MoS₂ layers of 39 ± 5 nm lateral size (20 wt % loading) creates interfacial contacts with relatively large number of MoS₂ edge sites and efficient electronic transport phenomena, yielding a high photocatalytic H₂‐production activity of 1497 μmol h⁻¹ gcat⁻¹ and an apparent QY of 3.3% at 410 nm light irradiation. This study thus offers a design strategy to improve light energy conversion efficiency of catalysts by engineering interfaces at the nanoscale in 2D‐layered heterojunction materials.
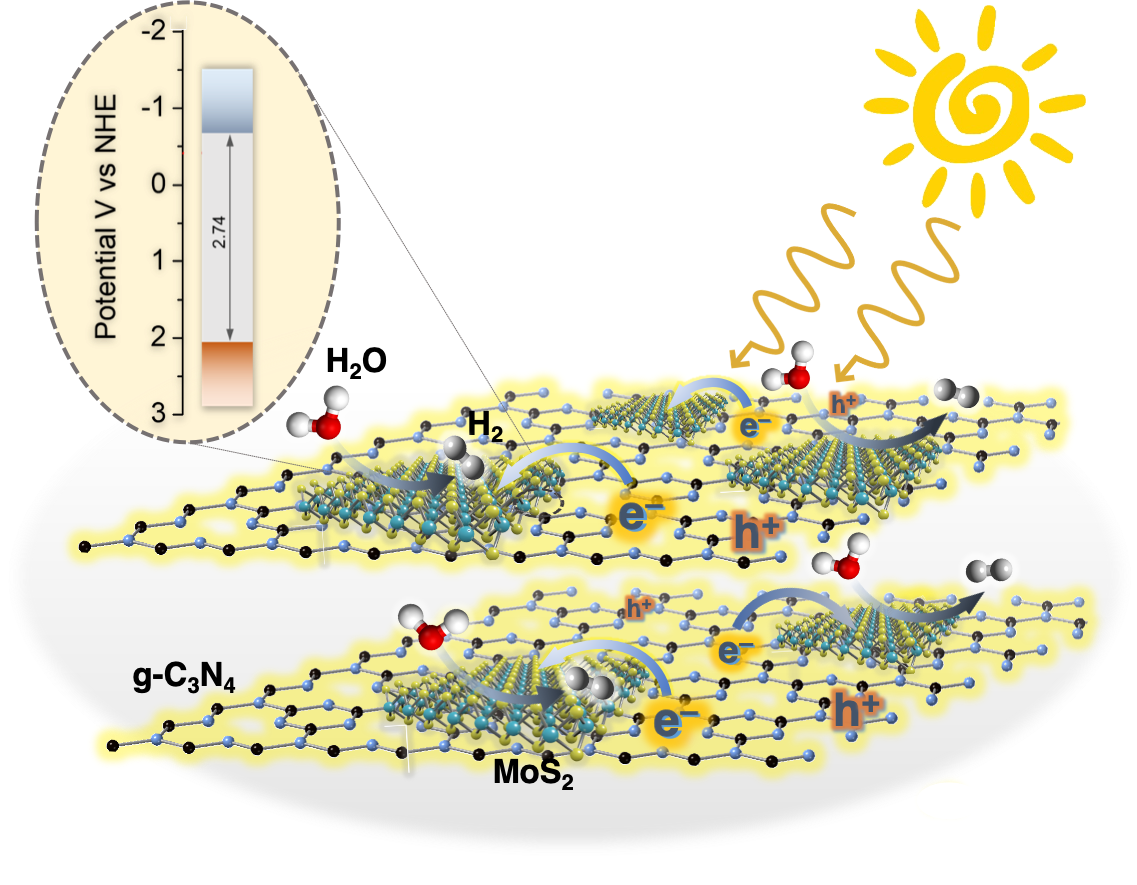
(see our Cover Picture, ChemPlusChem, 85, 1366, 2020)
(*This article also appears in Hot Topics: "Water Splitting", ChemElectroChem, "Photocatalysis", ChemPhotoChem, "Carbon, Graphite, and Graphene", Adv. Mater., 2020)
[104] I.T. Papadas, F. Galatopoulos, G.S. Armatas, N. Tessler & S.A. Choulis. Nanoparticulate Metal Oxide Top Electrode Interface Modification Improves the Thermal Stability of Inverted Perovskite Photovoltaics. Nanomaterials, 9, 1616–1623 (2019).
Solution processed γ-Fe₂O₃ nanoparticles via the solvothermal colloidal synthesis in conjunction with ligand-exchange method are used for interface modification of the top electrode in inverted perovskite solar cells. In comparison to more conventional top electrodes such as PC(70)BM/Al and PC(70)BM/AZO/Al, we show that incorporation of a γ-Fe₂O₃ provides an alternative solution processed top electrode (PC(70)BM/γ-Fe₂O₃/Al) that not only results in comparable power conversion efficiencies but also improved thermal stability of inverted perovskite photovoltaics. The origin of improved stability of inverted perovskite solar cells incorporating PC(70)BM/γ-Fe₂O₃/Al under accelerated heat lifetime conditions is attributed to the acidic surface nature of γ-Fe₂O₃ and reduced charge trapped density within PC(70)BM/γ-Fe₂O₃/Al top electrode interfaces.
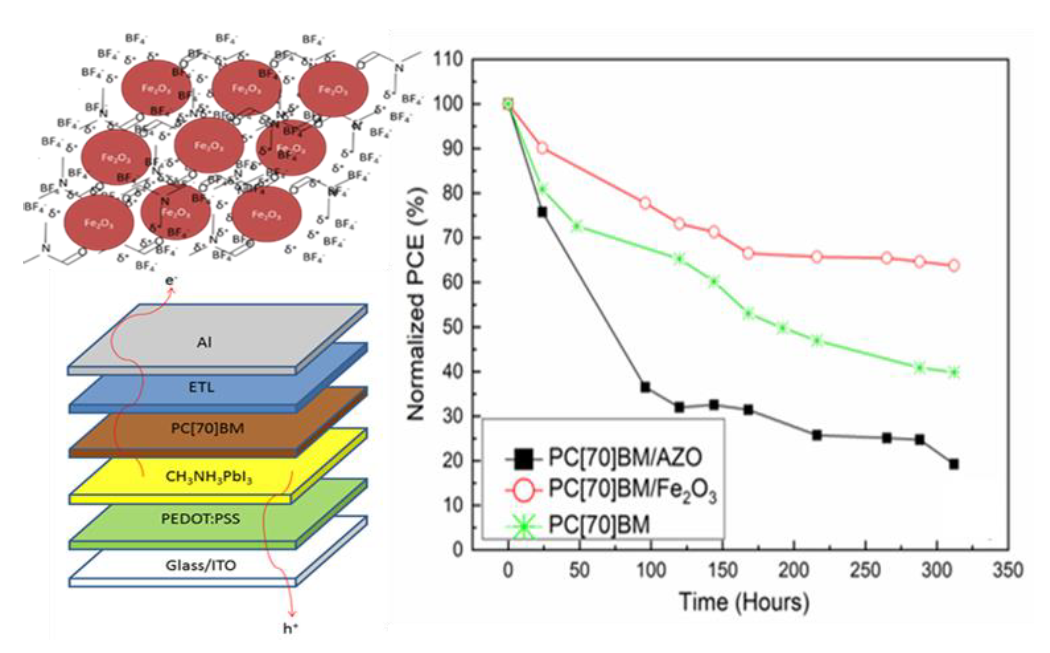
[103] A. Savva, I.T. Papadas, D. Tsikritzis, A. Ioakeimidis, F. Galatopoulos, K. Kapnisis, R. Fuhrer, B. Hartmeier, M.F. Oszajca, N.A. Luechinger, S. Kennou, G.S. Armatas & S.A. Choulis. Inverted Perovskite Photovoltaics using Flame Spray Pyrolysis Solution based CuAlO₂/Cu-O Hole Selective Contact. ACS Appl. Energy Mater., 2, 2276–2287 (2019).
We present the functionalization process of a conductive and transparent CuAlO₂/Cu-O hole transporting layer (HTL). The CuAlO₂/Cu-O powders were developed by flame spray pyrolysis and their stabilized dispersions were treated by sonication and centrifugation methods. We show that when the supernatant part of the treated CuAlO₂/Cu-O dispersions is used for the development of CuAlO₂/Cu-O HTLs the corresponding inverted perovskite–based solar cells show improved functionality and power conversion efficiency of up to 16.3% with negligible hysteresis effect.
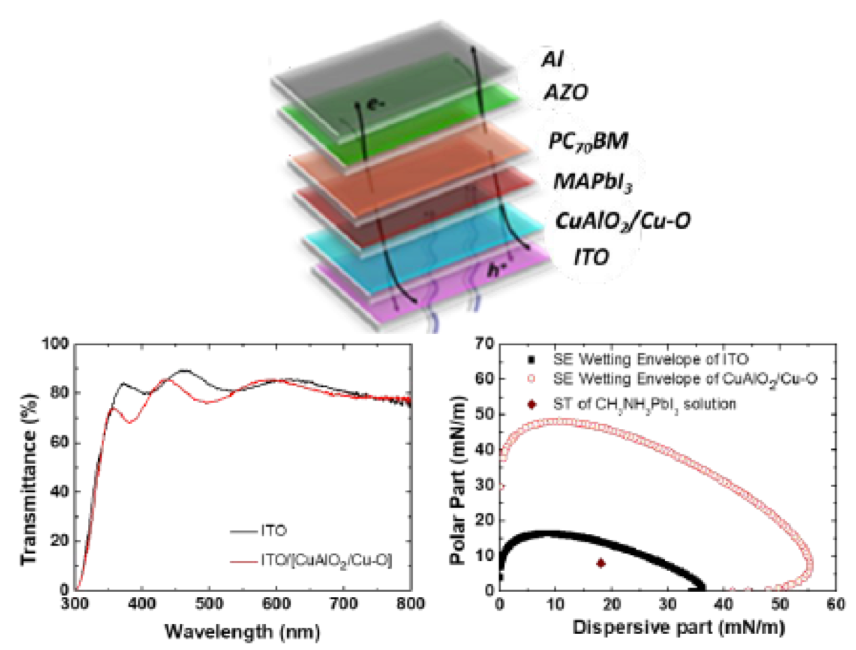
[102] Y. Georgiou, I.T. Papadas, E. Mouzourakis, E. Skliri, G.S. Armatas & Y. Deligiannakis. Mesoporous Spinel CoFe₂O₄ as Efficient Adsorbent for Arsenite Removal from Water: High Efficiency via Control of Particle Assemblage Configuration. Environ. Sci.: Nano, 6, 1156–1167 (2019).
A 3D CoFe₂O₄ mesoporous network of ~6 nm cobalt ferrite (CoFe₂O₄) nanoparticles (NPs), synthesized through a polymer-assisted aggregating self-assembly method, is presented. The single-phase crystallinity and high porosity of the obtained material were confirmed with X-ray diffraction, transition electron microscopy, Raman and N₂ porosimetry studies. Porous CoFe₂O₄ assemblies, obtained after heat treatment of the hybrid networks, possess an open-pore structure with a BET surface area of 160 m² g⁻¹ and pores with average size ~5.7 nm. Owing to its 3D network assemblage, this mesoporous CoFe₂O₄ exhibits an exceptional Asᴵᴵᴵ uptake capacity of 252.8 mg g⁻¹, which is much higher than that of random CoFe₂O₄ NP aggregates (47.3 mg g⁻¹) and bulk-like CoFe₂O₄ microparticles (43.6 mg g⁻¹). A comprehensive surface complexation model is presented, allowing a quantitative description of the Asᴵᴵᴵ adsorption on Fe and Co-sites. Our results indicate that Asᴵᴵᴵ uptake can be attributed to specific ≡FeOH and ≡CoOH sites located on the outer surface and interior pore voids of the material. Confinement inside the pores is found to be responsible for strong lateral interactions among the adsorbed [H₃AsO₃] species. The Asᴵᴵᴵ uptake of the present CoFe₂O₄ material is 3 to 10-fold higher than other high-performance adsorbents, such as graphite oxide, ZVI/activated carbon and CoFe₂O₄ and NiFe₂O₄ nanostructures. This exemplifies that, apart from surface chemistry, fine tuning of the spatial arrangement of NPs can offer advantageous tools towards highly-efficient Asᴵᴵᴵ adsorbents.
[101] A. Ioakeimidis, I.T. Papadas, D. Tsikritzis, G.S. Armatas, S. Kennou & S.A. Choulis. Enhanced Photovoltaic Performance of Perovskite Solar Cells by Co-Doped Spinel Nickel Cobaltite Hole Transporting Layer. APL Materials, 7, 021101 (2019).
A solution combustion synthesized hole transport layer (HTL) of spinel nickel cobaltite (NiCo₂O₄) incorporating 3% Cu–2% Li was fabricated using the doctor-blading technique for planar inverted perovskite solar cells (PVSCs). PVSCs incorporating 3% Cu–2% Li-doped NiCo₂O₄ showed an increase in Jsc and Voc device performance parameters compared to unmodified NiCo₂O₄, leading to power conversion efficiency (PCE) of 16.5%. X-ray photoelectron spectroscopy measurements revealed the tendency of Cu cations to replace preferably the surface Ni atoms by changing the surface stoichiometry of NiCo₂O₄, inducing a cathodic polarization. Ultraviolet photoelectron spectroscopy measurements unveiled the increase in the ionization potential by 0.1 eV for a co-doped NiCo₂O₄ film compared to unmodified NiCo₂O₄-based HTL. We attribute the enhanced PCE of the inverted PVSCs presented to the improved hole extraction properties of 3% Cu–2% Li NiCo₂O₄ HTL.
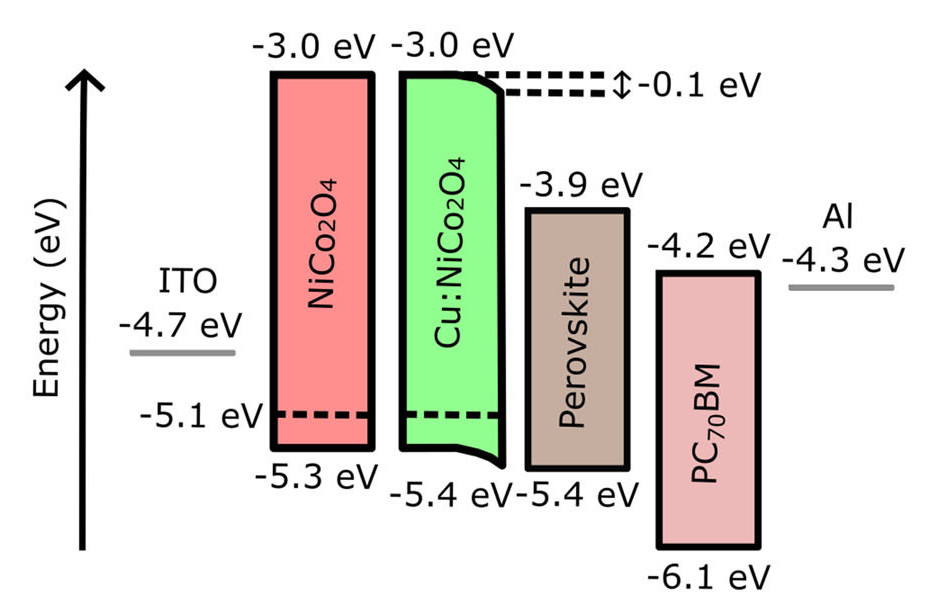
[100] G. Velegraki, I. Vamvasakis, I.T. Papadas, S. Tsatsos, A. Pournara, M.J. Manos, S. Choulis, S. Kennou, G. Kopidakis & G.S. Armatas. Boosting Photochemical Activity by Ni Doping of Mesoporous CoO Nanoparticle Assemblies. Inorg. Chem. Front., 6, 765–774 (2019).
The rational design of semiconductor nanostructures is of utmost importance for efficient solar energy conversion and environmental remediation. In this article, we report high-surface-area mesoporous networks consisting of Ni-implanted cubic CoO (Co1-xNixO) nanoparticles as promising catalysts for detoxification of Cr(VI) aqueous solutions. Mechanistic studies with X-ray photoelectron, UV–vis optical absorption, fluorescence and electrochemical impedance spectroscopy and theoretical (DFT) calculations indicate that the performance enhancement of these catalysts arises from the high charge transfer kinetics and oxidation efficiency of surface-reaching holes. By tuning chemical composition, the Co1-xNixO mesoporous catalyst at 2 atomic% Ni content imparts outstanding photocatalytic Cr(VI) reduction and water oxidation activity, corresponding to an apparent quantum yield (QY) of 1.5% at λ = 375 nm irradiation light.
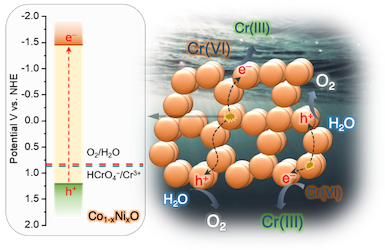
[99] S. Rapti, S. Diamantis, A. Dafnomili, A. Pournara, E. Skliri, G.S. Armatas, A. Tsipis, I. Spanopoulos, C.D. Malliakas, M.G. Kanatzidis, J.C. Plakatouras, N. Fotini, T. Lazarides & M.J. Manos. Exceptional TcO₄⁻ sorption capacity and highly efficient ReO₄⁻ luminescence sensing by Zr₄⁺ MOFs. J. Mater. Chem. A, 6, 20813–20821 (2018).
The sorption properties of [Zr₆O₄(OH)₄(NH₃⁺-BDC)₆]Cl₆∙xH₂O (MOR-1) and H₁₆[Zr₆O₁₆(H₂PATP)₄]Cl₈∙xH₂O (MOR-2) towards ReO₄⁻ and TcO₄⁻ were studied in detail. Both MOR-1 and MOR-2 are very effective sorbents for ReO₄⁻ and TcO₄⁻ anions, with MOR-2 showing the highest sorption capacity (up to 4.1 ±0.4 mmol/g) among known metal organic materials. Importantly, the exceptional sorption capacity of MOR-2 is retained even under conditions simulating acidic nuclear waste. In addition, MOR-1 and MOR-2 exchibit selective luminescence ReO₄⁻ sensing properties, first time, demonstrated for MOF materials.
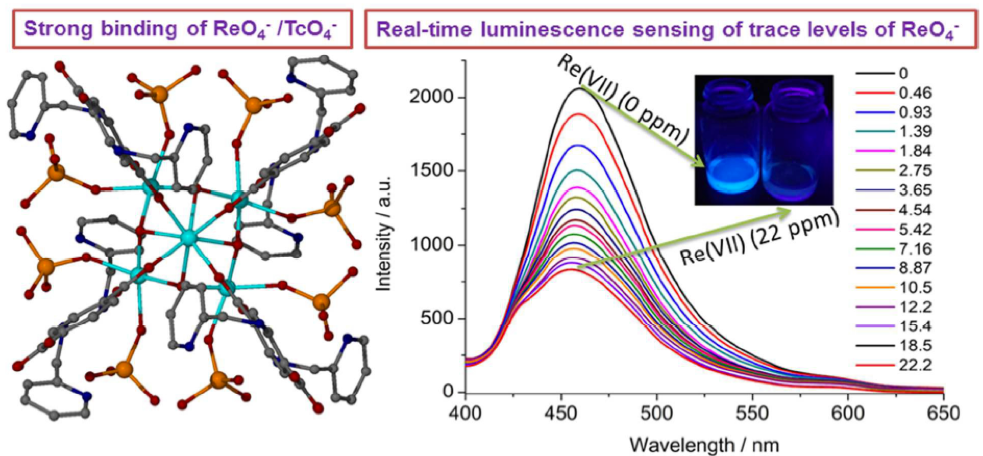
[98] I. Vamvasakis, I.T. Papadas, Th. Tzanoudakis, Ch. Drivas, S.A. Choulis, S. Kennou & G.S. Armatas. Visible-Light Photocatalytic H₂ Production Activity of β-Ni(OH)₂ Modified CdS Mesoporous Nano-Heterojunction Networks. ACS Catal., 8, 8726–8738 (2018).
Photocatalytic water splitting for hydrogen production is an emerging and promising strategy for converting solar energy into chemical fuels. To that end, the development of robust and highly active semiconductor materials is of eminent importance in this field. Here, we demonstrate high-surface-area mesoporous networks comprising interconnected β-Ni(OH)₂ modified CdS nanocrystals (NCs) as highly active and stable photocatalysts for hydrogen generation. Compared to single-component CdS assemblies, Ni-modified materials present a strong enhancement of photocatalytic performance for hydrogen evolution under visible light irradiation (λ ≧ 420 nm). By controlling the formation of β-Ni(OH)₂ species, the mesoporous β-Ni(OH)₂/CdS heterojunction networks at a 10 wt % Ni content reached an outstanding photocatalytic H₂ evolution rate of 1.4 mmol h⁻¹ at 20 ∘C (or ~35 mmol g⁻¹ h⁻¹ mass activity), associated with an apparent quantum yield (QY) of 72% at 420 nm in a 5 M NaOH aqueous solution containing 10% v/v ethanol as sacrificial reagent. Mechanistic study with UV–vis/NIR, PL and EIS spectroscopy and photocatalytic performance evaluation reveals that the improved photocatalytic performance arises from the strong electronic coupling and charge-transferred states at the p–n β-Ni(OH)₂/CdS heterojunctions. These β-Ni(OH)₂ modified CdS mesoporous assemblies have important implications for renewable hydrogen generation technologies.
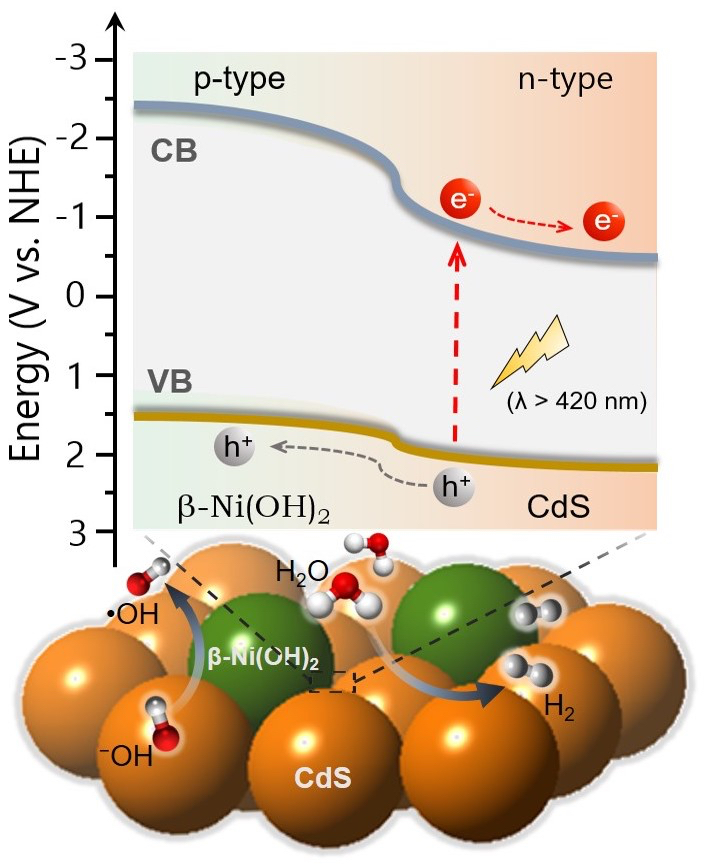
[97] F. Galatopoulos. I.T. Papadas, G.S. Armatas & S.A. Choulis. Long Thermal Stability of Inverted Perovskite Photovoltaics Incorporating Fullerene-Based Blocking Layer. Adv. Mater. Interfaces, 5, 1800270 (2018).
In this article, the stability of inverted (p‐i‐n) perovskite solar cells is studied under accelerated heat lifetime conditions (60 °C, 85 °C, and N₂ atmosphere). By using a combination of buffer layer engineering, impedance spectroscopy, and other characterization techniques, the interaction of the perovskite active layer with the top Al metal electrode through diffusion mechanisms is proposed as the major thermal degradation pathway for planar inverted perovskite photovoltaics (PVs) under 85 °C heat conditions. It is shown that by using thick solution processed fullerene buffer layer the perovskite active layer can be isolated from the top metal electrode and improve the lifetime performance of the inverted perovskite photovoltaics at 85 °C. Finally, an optimized reliable solution processed inverted perovskite PV device using thick fullerene diffusion blocking layer with over 1000 h accelerated heat lifetime performance at 60 °C is presented.
[96] I. Tamiolakis, D. Liu, F.-X. Xiao, J. Xie, I.T. Papadas, T. Salim, B. Liu, Q. Zhang, S.A. Choulis & G.S. Armatas. Mesoporous Implantable Pt/SrTiO₃:C,N Nanocuboids Delivering Enhanced Photocatalytic H₂-Production Activity via Plasmon-Induced Interfacial Electron Transfer. Appl. Catal. B: Environ., 236, 338–347 (2018).
Band edge engineering of semiconductor nanostructures is one of the most appealing approaches to enhancelight absorption, carrier separation and, ultimately, solar to fuel conversion efficiency. In this study, we devise a facile polymer-assisted sol-gel chemical method to prepare highly porous, crystalline implanted SrTiO₃ (STO) nanoparticles and demonstrate their performance for photocatalytic hydrogen generation from water. X-ray scattering, electron microscopy, and nitrogen physisorption data corroborate that the as-made catalysts comprise 100-nm-sized nanocuboid particles containing a highly internal porous structure (BET surface area ∼176 m² g⁻¹) with uniform mesopores (ca. 5.8 nm in diameter). Interestingly, a partial substitution of N and C for O is attained in STO lattice with this synthetic protocol, according to the elemental analysis, and infrared (IR) and Xray photoelectron spectroscopy (XPS) studies. Compared to STO:C,N, the STO:C,N mesoporous decorated with Pt nanoparticles (ca. 3 nm) present unique attributes that allow for an impressive improvement of up to 74-fold in photocatalytic H₂-production activity. By combining UV–vis/NIR optical absorption, photoluminescence, Raman and electrochemical impedance spectroscopy, we show that this improved performance arises from the unique nanostructure, which provides massive surface active sites, and the proper alignment of defect states and conduction band-edge position of the STO:C,N semiconductor with respect to the interband transitions of metal, which permit efficient plasmon-induced interfacial electron transfer between the Pt–STO:C,N junction.
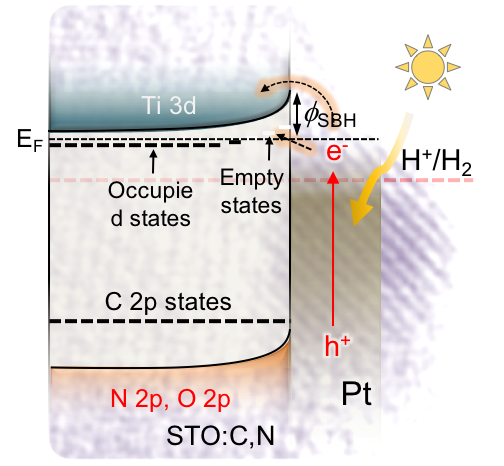
[95] I.T. Papadas, A. Savva, A. Ioakeimidis, P. Eleftheriou, G.S. Armatas & S.A. Choulis. Employing Surfactant-Assisted Hydrothermal Synthesis to Control CuGaO₂ Nanoparticle Formation and Improved Carrier Selectivity of Perovskite Solar Cells. Mater. Today Energy, 8, 57–64 (2018).
Delafossites like CuGaO₂ have appeared as promising p-type semiconductor materials for opto-electronic applications mainly due to their high optical transparency and electrical conductivity. However, existing synthetic efforts usually result in particles with large diameter limiting their performance relevant to functional electronic applications. In this article, we report a novel surfactant-assisted hydrothermal synthesis method, which allows the development of ultrafine (∼5 nm) monodispersed p-type CuGaO₂ nanoparticles (NPs). We show that DMSO can be used as a ligand and dispersing solvent for stabilizing the CuGaO₂ NPs. The resulting dispersion is used for the fabrication of dense, compact functional CuGaO2 electronic layer with properties relevant to advanced optoelectronic applications. As a proof of concept, the surfactant-assisted hydrothermal synthesized CuGaO₂ is incorporated as a hole transporting layer (HTL) in the inverted p-i-n perovskite solar cell device architecture providing improved hole carrier selectivity and power conversion efficiency compared to conventional PEDOT:PSS HTL based perovskite solar cells.
[94] I.T. Papadas, A. Ioakeimidis, G.S. Armatas & S.A. Choulis. Low Temperature Combustion Synthesis of a Spinel NiCo₂O₄ Hole Transport Layer for Perovskite Photovoltaics. Adv. Sci., 5, 1701029 (2018).
The synthesis and characterization of low‐temperature solution‐processable monodispersed nickel cobaltite (NiCo₂O₄) nanoparticles (NPs) via a combustion synthesis is reported using tartaric acid as fuel and the performance as a hole transport layer (HTL) for perovskite solar cells (PVSCs) is demonstrated. NiCo₂O₄ is a p‐type semiconductor consisting of environmentally friendly, abundant elements and higher conductivity compared to NiO. It is shown that the combustion synthesis of spinel NiCo₂O₄ using tartaric acid as fuel can be used to control the NPs size and provide smooth, compact, and homogeneous functional HTLs processed by blade coating. Study of PVSCs with different NiCo₂O₄ thickness as HTL reveals a difference on hole extraction efficiency, and for 15 nm, optimized thickness enhanced hole carrier collection is achieved. As a result, p‐i‐n structure of PVSCs with 15 nm NiCo₂O₄ HTLs shows reliable performance and power conversion efficiency values in the range of 15.5% with negligible hysteresis.

[93] E. Skliri, J. Miao, J. Xie, G. Liu, T. Salim, B. Liu, Q. Zhang & G.S. Armatas. Assembly and Photochemical Properties of Mesoporous Networks of Spinel Ferrite Nanoparticles for Environmental Photocatalytic Remediation. Appl. Catal. B: Environ., 227, 330–339 (2018).
Spinel ferrite materials have an electronic band structure that is well suited for visible light-induced catalysis, however, their photocatalytic activity remains low due to the daunting charge-carrier separation process. In this article, we report that high-surface-area mesoscopic architectures composed of tightly connected ultrasmall spinel ferrite nanocrystals can efficiently suppress electron-hole recombination, manifesting an exceptional activity and magnetic recyclability in photocatalytic reduction of aqueous Cr(VI). Revealed by electron microscopy, N₂ physisorption, and X-ray scattering studies, the resulting materials, which were obtained through a block copolymer-assisted cross-linking aggregation of colloidal nanoparticles, show a 3D interconnected nanoporous structure with a large internal surface area (up to 159 m² g⁻¹) and exhibit small grain composition (ca. 6–8 nm in size). Through a systematic synthesis of various structural analogues to the spinel ferrite family and optical absorption and electrochemical impedance spectroscopy analyses, we demonstrate that the electronic band structure fits the electronic requirements for both Cr(VI) reduction and water oxidation under UV–vis light irradiation. Among spinel ferrites, ZnFe₂O₄ presents the highest activity, readily operating without additional sacrificial reagents in photocatalytic detoxification of aqueous Cr(VI), which together with transient gas analysis and fluorescence spectroscopy results suggest a competitive formation of oxygen and hydroxyl radicals at the catalyst surface. These findings provide an essential tool for the delineation of the electronic structure-catalytic property relationship in spinel ferrite nanostructures offering intriguing possibilities for designing new photocatalytic systems for efficient environmental pollution purification and energy conversion.
[92] P. Karaolia, I. Michael-Kordatou, E. Hapeshi, C. Drosou, Y. Bertakis, D. Christofilos, G.S. Armatas, L. Sygellou, T. Schwartz, N.P. Xekoukoulotakis & D. Fatta-Kassinos. Removal of antibiotics, antibiotic-resistant bacteria and their associated genes by graphene-based TiO₂ composite photocatalysts under solar radiation in urban wastewaters. Appl. Catal. B: Environ., 224, 810–824 (2018).
The present work investigated: (i) the removal of the antibiotics sulfamethoxazole (SMX), erythromycin (ERY) and clarithromycin (CLA); (ii) the inactivation of the total and antibiotic-resistant E. coli along with their regrowth potential after treatment; (iii) the removal of the total genomic DNA content; and (iv) the removal of selected antibiotic resistance genes (ARGs), namely sul1, ampC, ermB, mecA, as well as species-specific sequences, namely ecfX for Pseudomonas aeruginosa and enterococci-specific 23S rRNA, by graphene-based TiO₂ composite photocatalysts under solar radiation, in real urban wastewaters. TiO2-reduced graphene oxide (TiO2-rGO) composite photocatalysts were synthesized by two ex-situ synthesis methods, namely hydrothermal (HD) treatment and photocatalytic (PH) treatment, starting from graphene oxide and Aeroxide P25 TiO₂, and were characterized with various techniques, such as XRD, FT-IR, Raman, XPS, SEM and surface area (BET) analyses. The potential of the synthesized TiO₂-rGO composites for the removal of the abovementioned antibiotic-related microcontaminants was compared to the efficiency shown by pristine Aeroxide P25 TiO₂ under simulated solar radiation, in real urban wastewater effluents treated by a membrane bioreactor. The results showed that TiO2-rGO-PH was more efficient in the photocatalytic degradation of ERY (84 ± 2%) and CLA (86 ± 5%), while degradation of SMX (87 ± 4%) was found to be slightly higher with Aeroxide P25 TiO₂. It was also demonstrated that more than 180 min of treatment were satisfactory for the complete inactivation and complete absence of post-treatment regrowth of E. coli bacteria (LOD) even 24 h after the end of the treatment, for all examined photocatalytic materials. The least amount of regrowth at all experimental times was observed in the presence of TiO₂-rGO-HD. Moreover, the synthesized graphene-based photocatalysts successfully removed ampC and significantly reduced ecfX abundance of Pseudomonas aeruginosa, but sul1, ermB and 23S rRNA for enterococci sequences were found to be persistent throughout treatment with all catalyst types. Finally, the total DNA concentration remained stable throughout the photocatalytic treatment (4.2–4.8 ng μL⁻¹), indicating the high total genomic DNA stability in treated wastewater and its resistance to photocatalytic treatment.
[91] G. Velegraki, J. Miao, Ch. Drivas, B. Liu, S. Kennou & G.S. Armatas. Fabrication of 3D Mesoporous Networks of Assembled CoO Nanoparticles for Efficient Photocatalytic Reduction of Aqueous Cr(VI). Appl. Catal. B: Environ., 221, 635–644 (2018).
Synthesis of high-performance and cyclic stable photocatalysts has been remaining a significant challenge. In this work, we report the synthesis of high-surface-area mesoporous networks of CoO NPs through a polymer-templating self-assembly method and demonstrate their potential application in the reductive detoxification of aqueous Cr(VI) solutions under UV and visible light irradiation. Electron microscopy images and N₂ adsorption measurements corroborate the presence of a porous network of interconnected CoO NPs (ca. 18 nm in size) with large internal surface area (up to 134 m² g⁻¹) and narrow pore-size distribution (ca. 4.4–4.8 nm in diameter). Conjunction of optical absorption and electrochemical impendence spectroscopy results indicates that the band edge positions of constituent CoO NPs meet the electric potential requirements for reducing Cr(VI) and splitting water to oxygen. We show that mesoporous assemblies of hexagonal CoO NPs effectively overcome the kinetic barriers for the oxidation reaction, manifesting a remarkably photocatalytic Cr(VI) reduction activity at acidic pH with an apparent quantum yield (AQY) of 1.61% and 0.17% at wavelengths of 375 and 440 nm, respectively. We demonstrate that, apart from oxygen evolution reaction, photoconversion of harmful Cr(VI) into non-toxic Cr(III) involves also a hydroxyl radical-mediated oxidation process by intercepting oxidation products with on-line mass spectrometry and fluorescence spectroscopy in control catalytic experiments.
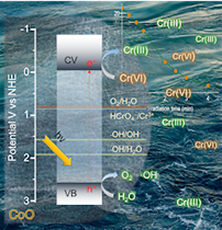
[90] G. Velegraki, J. Xie, Q. Zhang & G.S. Armatas. Mesoporous Copper Nanoparticle Networks Decorated by Graphite Layers for Surface-Enhanced Raman Scattering Detection of Trace Analytes. ChemPlusChem, 82, 1290–1297 (2017).
The assembly of 3D nanoscale structures of plasmonic nanoparticles (NPs) holds great promise for achieving enhanced optical and electronic properties. This type of materials exhibits a large number of surface hot spots, while offering the possibility for synergetic effects to be observed. Herein, a facile, yet powerful, strategy to fabricate 3D mesoporous networks of copper NPs decorated with graphite layers (denoted as Cu/G) is demonstrated by using a polymer‐assisted self‐assembly method. After thermal processing, the resulting Cu/G‐linked networks retain an open and interconnected porosity with a large surface area (up to 90 m² g⁻¹) and narrow pore size distribution (ca. 4.3 nm in size). Owing to these characteristics, Cu/G assemblies behave as high‐performance surface‐enhanced Raman scattering (SERS) probes for the detection of analytes in very low concentrations. The substrates comprise low‐cost, environmentally benign materials and show promise for chemical and biological sensing applications.
[89] A. Savva, I.T. Papadas, D. Tsikritzis, G.S. Armatas, S. Kennou & S. Choulis. Room Temperature Nanoparticulate Interfacial Layers for Perovskite Solar Cells via Solvothermal Synthesis. J. Mater. Chem. A, 5, 20381–20389 (2017).
We present a solvothermal synthetic route to produce monodisperse CuO nanoparticles (NPs) in the range of 5–10 nm that can be used as a hole selective interfacial layer between indium tin oxide (ITO) and the perovskite active layer for p–i–n perovskite solar cells by spin casting the dispersions at room temperature. The bottom electrode interface modification provided by spherical CuO-NPs at room temperature promotes the formation of high quality perovskite photoactive layers with a large crystal size and strong optical absorption. Furthermore, it is shown that the nanoparticulate nature of the CuO hole transporting interfacial layer can be used to improve light manipulation within the perovskite solar cell device structure. The corresponding p–i–n CH3NH3PbI3-based solar cells show high Voc values of 1.09 V, which is significantly higher compared to the Voc values obtained with conventional PEDOT:PSS hole selective contact based perovskite solar cells.

[88] A.D. Pournara, S. Rapti, E. Skliri, G.S. Armatas, A.C. Tsipis, & M.J. Manos. Highly Efficient Sorption of Methyl Orange by a Metal Organic Resin–Alginic Acid Composite. ChemPlusChem, 82, 1188–1196 (2017).
The composite anion‐exchange material MOR‐1–HA (metal–organic resin‐1–alginic acid) was investigated as sorbent for the capture of the methyl orange anion (MO−) from aqueous solutions. MOR‐1–HA shows a remarkably high sorption capacity (up to 859 mg g−1) and rapid sorption kinetics, the fastest among the reported metal–organic sorbents. It is capable of absorbing MO− over a wide pH range (1–8) and, in addition, it exhibits significant MO− sorption affinity even in the presence of large excesses of competing anions (e.g., Cl−, NO3−, SO42−). The exceptional MO−‐sorption properties of MOR‐1–HA arise not only from its highly porous structure and easily exchangeable Cl− anions, but also from a multitude of interaction effects, such as electrostatic interactions between MO− and the NH3+ groups of the material, hydration/dehydration, hydrophobicity/hydrophilicity, size and capacity of generating lateral interactions, and intercalation as revealed by theoretical studies. An ion‐exchange column with a stationary phase containing MOR‐1–HA and silica sand showed high efficiency for the removal of MO− from various types of aqueous samples. The column can be readily regenerated and reused for many runs with minimal loss (2.3–9.3 %) of its exchange capacity. The simplicity of the MOR‐1–HA/sand column and its high regeneration capability and reusability make it particularly attractive for application in the remediation of MO−‐contaminated industrial wastewater.
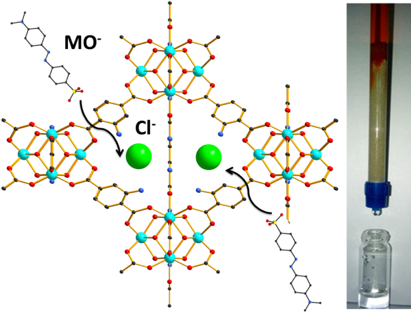
(*This article is part of the "Early Career Series", ChemPlusChem, 2017)
[87] S. Rapti, D. Sarma, S. A. Diamantis, E. Skliri, G.S. Armatas, A.C. Tsipis, Y.S. Hassan, M. Alkordi, C.D. Malliakas, M.G. Kanatzidis, T. Lazarides, J.C. Plakatouras & M.J. Manos. All in One Porous Material: Exceptional Sorption and Selective Sensing of Hexavalent Chromium by a Zr4+ MOF. J. Mater. Chem. A, 5, 14707–14719 (2017).
We report a new microporous metal–organic framework (MOF) H16[Zr6O16(H2PATP)4]Cl8·xH2O (H2PATP = 2-((pyridin-1-ium-2-ylmethyl)ammonio)terephthalate), denoted as MOR-2 (metal organic resin-2). MOR-2 represents the first Zr4+-terephthalate MOF with an 8-connected net and also the first example where a bulky functional group was introduced into the terephthalate scaffold prior to the MOF synthesis. MOR-2 shows extraordinary capability to rapidly capture (within 1 min) hexavalent chromium with a sorption capacity up to 194 mg Cr(VI) per g, which far exceeds those reported for the known Cr(VI) sorbents. Moreover, MOR-2 in its composite form with alginic acid (HA) can be utilized in ion exchange columns, which are highly efficient for the removal of Cr(VI) from aqueous solutions including industrial waste samples and also can be regenerated and reused several times with minimal loss (less than 20%) of their capacity. Besides an excellent sorbent, MOR-2 is also a highly efficient sensor for real time detection of Cr(VI) species as revealed by fluorescence titration experiments in acidic aqueous media. The Cr(VI) detection limits were found as low as 4 ppb, while the system exhibited excellent sensitivity when real world, instead of standard, samples were employed. Thus, the MOR-2 material is a unique example combining both excellent sorption and exceptional luminescence sensing of Cr(VI) species in aqueous solutions.
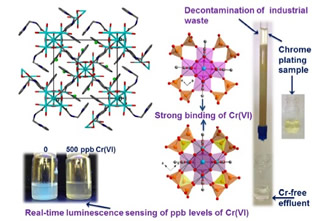
[86] I.U. Arachchige, G.S. Armatas, K. Biswas, K. Subrahmanyam, S. Latturner, C.D. Malliakas, M.J. Manos, Y. Oh, K. Polychronopoulou, P.F.P. Poudeu, P.N. Trikalitis, Q. Zhang, L.-D. Zhao & S.C. Peter. Mercouri G. Kanatzidis: Excellence and Innovations in Inorganic and Solid-State Chemistry. Inorg. Chem., 56, 7582–7597 (2017).
Over the last 3–4 decades, solid-state chemistry has emerged as the forefront of materials design and development. The field has revolutionized into a multidisciplinary subject and matured with a scope of new synthetic strategies, new challenges, and opportunities. Understanding the structure is very crucial in the design of appropriate materials for desired applications. Professor Mercouri G. Kanatzidis has encountered both challenges and opportunities during the course of the discovery of many novel materials. Throughout his scientific career, Mercouri and his group discovered several inorganic compounds and pioneered structure–property relationships. We, a few Ph.D. and postdoctoral students, celebrate his 60th birthday by providing a Viewpoint summarizing his contributions to inorganic solid-state chemistry. The topics discussed here are of significant interest to various scientific communities ranging from condensed matter to green energy production.
[85] Q. Zhang, G.S. Armatas & J. Aitken. Mercouri G. Kanatzidis. Thirty Years of Contributions to Materials and Inorganic Chemistry. Inorg. Chem. Front., 4, 1098–1099 (2017).
It is our great honor to write this Editorial for the themed collection in honour of Professor Mercouri G. Kanatzidis for his scientific contributions to materials and inorganic chemistry over the past 30 years, as well as to mark the occasion of his 60th birthday. Mercouri, as one of the leading scientists adopting revolutionary pathways and innovative ideas for shaping and advancing the field of materials and solid-state chemistry, has many remarkable achievements in the fields of synthetic inorganic chemistry, exploratory synthesis and the development of new functional materials, including perovskite materials with applications in solar cells, thermoelectrics, nanostructured chalcogenide materials, complex intermetallic phases, and superconductors.


(*This article is part of the Thematic Issue: "In honour of Mercouri G. Kanatzidis for his contributions to Inorganic Chemistry for over 30 years", Inorg. Chem. Front., 2017)
[84] G. Liu, J. Liu, L. Nie, R. Ban, G.S. Armatas, X. Tao & Q. Zhang. Surfactant 1-Hexadecyl-3-methylimidazolium Chloride Can Convert One-Dimensional Viologen Bromoplumbate into Zero-Dimensional. Inorg. Chem., 56, 5498–5501 (2017).
A zero-dimensional N,N′-dibutyl-4,4′-dipyridinium bromoplumbate, [BV]6[Pb9Br30], with unusual discrete [Pb9Br30]12– anionic clusters was prepared via a facile surfactant-mediated solvothermal process. This bromoplumbate exhibits a narrower optical band gap relative to the congeneric one-dimensional viologen bromoplumbates.
[83] L. Nie, G. Liu, J. Xie, T.-T. Lim, G.S. Armatas, R. Xu & Q. Zhang. Syntheses, Crystal Structures, and Photocatalytic Properties of Two Ammonium-Directed Ag−Sb−S Complexes. Inorg. Chem. Front., 4, 954–959 (2017).
With ammonium ions as structure-directing agents, two novel silver thioantimonates (NH4)AgSb4S7·H2O (1) and (NH4)AgSb2S4 (2) have been surfactant-thermally synthesized and fully characterized. Compound 1 features a three dimensional (3D) open framework while compound 2 possesses a two dimensional (2D) layer structure. The optical band gaps are estimated to be 1.70 eV for 1 and 1.84 eV for 2, exhibiting semiconductor properties for visible light absorption. The photocatalytic experiments demonstrated that both 1 and 2 were able to degrade crystal violet (CV) and rhodamine B (RhB) under visible light irradiation. Moreover, compounds 1 and 2 also displayed selective photocatalytic abilities in CV degradation.
[82] E.-E. Vlachou, G.S. Armatas & K.E. Litinas. Synthesis of Fused Oxazolocoumarins from o-Hydroxynitrocoumarins and Benzyl Alcohol Under Gold Nanoparticles or FeCl3 Catalysis. J. Heterocyclic Chem., 54, 2447–2453 (2017).
Synthesis of fused oxazolocoumarins has been achieved from the one‐pot tandem reactions of o‐hydroxynitrocoumarins with benzyl alcohol in toluene under catalysis in a sealed tube at 150°C. The catalysis was performed by gold nanoparticles supported on TiO2 (0.4 mol% Au) or FeCl3 (5%) or silver nanoparticles supported on TiO2 (1.7 mol% Ag).

[81] E. Papazoi, A. Douvali, S. Rapti, E. Skliri, G.S. Armatas, G.S. Papaefstathiou, X. Wang, Z. Huang , S. Kaziannis, C. Kosmidis, A. Hatzidimitriou, T. Lazarides & M. Manos. A Microporous Mg2+ MOF with High Selectivity for CO2 and Cation Exchange Property in a Single-Crystal-to-Single-Crystal Fashion. Inorg. Chem. Front., 4, 530–536 (2017).
We report herein a new alkaline earth metal ion organic framework [Mg2(NH2BDC)2(HNO3)]·9H2O (AEMOF-7), which shows a 3-D microporous structure with several unusual features, such as the rare trigonal prismatic coordination geometry of one of the crystallographically unique Mg2+ centers and the existence of a bridging HNO3 ligand. The H+ ions of the HNO3 ligand are dissociable as demonstrated via proton conductivity measurements. AEMOF-7 displays relatively high selectivity for CO2vs. CH4 and negligible N2 uptake. Interestingly, this compound was found to be capable of single-crystal-to-single-crystal (SCSC) exchange of Mg2+ by Cu2+ ions, which was observed for the first time in a MOF material. AEMOF-7 is also luminescent and its photophysical properties were investigated via solid state UV-Vis, steady-state and time-resolved luminescence studies.
(*This article is part of the Thematic Issue: "In honour of Mercouri G. Kanatzidis for his contributions to Inorganic Chemistry for over 30 years", Inorg. Chem. Front., 2017)
[80] I. Vamvasakis, A. Trapali, J. Miao, B. Liu & G.S. Armatas. Enhanced Visible-Light Photocatalytic Hydrogen Production Activity of Three-Dimensional Mesoporous p-CuS/n-CdS Nanocrystal Assemblies. Inorg. Chem. Front., 4, 433–441 (2017).
Transition metal sulfide nanoparticles currently attract enormous research attention because of their high photon-to-electron conversion efficiency, excellent electron conductivity and low cost. However, single-component metal sulfides suffer from low charge-carrier separation yield and poor chemical stability. Here, we report the synthesis of mesoporous structures of n-type CdS and p-type CuS nanocrystals and investigate their photocatalytic performance for the generation of hydrogen from water. X-ray diffraction, transmission electron microscopy and nitrogen physisorption data indicate that the resultant CuS/CdS heterostructures consist of a highly porous network (BET surface area ∼198–203 m2 g−1) of connected 5 nm-sized nanoparticles and contain uniform mesopores (ca. 6.4 nm in diameter) within the assembled structure. The nanoparticles are composed of CuS compounds which are in intimate contact with CdS nanocrystals, and these results are corroborated with energy-dispersive and X-ray photoelectron spectroscopy measurements. These CuS/CdS binary semiconductors can carry out photocatalytic reduction of water under visible light irradiation with a quantum efficiency up to ∼12.6% at a wavelength of 420 ± 10 nm. UV–vis/NIR, photoluminescence and electrochemical impedance spectroscopy studies show that the superior photocatalytic activity of the CuS/CdS nanoparticle networks mainly arises from a proper alignment of the band-edge positions of the materials, which suppress carrier recombination and permit efficient interfacial charge transport at the p-CuS/n-CdS junction region.
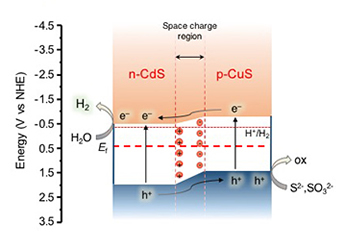
(*This article is part of the Thematic Issue: "In honour of Mercouri G. Kanatzidis for his contributions to Inorganic Chemistry for over 30 years", Inorg. Chem. Front., 2017)
[79] E. Skliri, S. Papadogiorgakis, I.N. Lykakis & G.S. Armatas. Mesoporous Assembled Mn3O4 Nanoparticle Networks as Efficient Catalysts for Selective Oxidation of Alkenes and Aryl Alkanes. ChemPlusChem, 82, 136–143 (2017).
The design of nanoscale materials has been considered important for enhancing their surface properties for catalysis. Metal oxide nanoparticles have a large number of exposed surface active sites, but they suffer from low reactivity and poor stability resulting from excessive aggregation into less active microscopic structures. Herein, the synthesis of mesoporous Mn3O4 nanoparticle assemblies by polymer‐assisted self‐assembly is presented and their catalytic activity is demonstrated in the oxidation of various saturated and unsaturated hydrocarbons, including aromatic alkenes and aryl alkanes, in the presence of tert‐butyl hydroperoxide as a mild oxidant. It is also shown through comparative studies that the high catalytic activity and stability of these Mn3O4 assemblies arise from the unique three‐dimensional open‐pore structure, high internal surface area (90 m2 g−1) and uniform mesopores (≈6.6 nm in size).
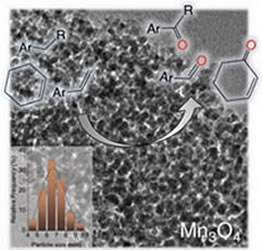
(*Highlighted by ChemistryViews.org, Nanoparticle Networks for Improved Catalysis, 2016)
(*This article is part of the Special Issue: "5th Anniversary Virtual Issue", ChemPlusChem, 2017)
[78] I. Vamvasakis, B. Liu & G.S. Armatas. Size Effects of Platinum Nanoparticles in the Photocatalytic Hydrogen Production over 3D Mesoporous Networks of CdS and Pt Nanojunctions. Adv. Funct. Mater., 26, 8062–8071 (2016).
Catalysts for the photogeneration of hydrogen from water are key for realizing solar energy conversion. Despite tremendous efforts, developing hydrogen evolution catalysts with high activity and long‐term stability remains a daunting challenge. Herein, the design and fabrication of mesoporous Pt‐decorated CdS nanocrystal assemblies (NCAs) are reported, and their excellent performance for the photocatalytic hydrogen production is demonstrated. These materials comprise varying particle size of Pt (ranging from 1.8 to 3.3 nm) and exhibit 3D nanoscale pore structure within the assembled network. Photocatalytic measurements coupled with UV–vis/NIR optical absorption, photoluminescence, and electrochemical impedance spectroscopy studies suggest that the performance enhancement of these catalytic systems arises from the efficient hole transport at the CdS/electrolyte interface and interparticle Pt/CdS electron‐transfer process as a result of the deposition of Pt. It is found that the Pt‐CdS NCAs catalyst at 5 wt% Pt loading content exerts a 1.2 mmol h−1 H2‐evolution rate under visible‐light irradiation (λ ≥ 420 nm) with an apparent quantum yield of over 70% at wavelength λ = 420 nm in alkaline solution (5 m NaOH), using ethanol (10% v/v) as sacrificial agent. This activity far exceeds those of the single CdS and binary noble metal/CdS systems, demonstrating the potential for practical photocatalytic hydrogen production.
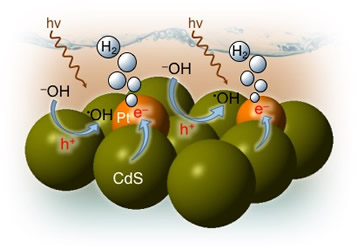
[77] A.G. Margellou, I.T. Papadas, D.E. Petrakis & G.S. Armatas. Development of Enhanced Surface Area LaFeO3 Perovskites Using Amino Acids as Templating Agents. Mater. Res. Bull., 83, 491–501 (2016).
In this article LaFeO3 perovskites of enhanced specific surface area (ssa) have been developed through a soft templating path which involves amino acids as capping agents of primary particles, in order to suppress aggregating effects, and polymer P123 as a surface enhancing agent. The treatment of the primary LaFeO3 particles took place using the amino acids β-alanine, serine, tyrosine and threonine and the final surface area increased from 18 up to 42⿿59 m2/g, depending on the amino acid. The obtained material showed also a remarkable red shift band gap. The main factor affecting the development of ssa seems to be the interactions between the solid particles and the amphiphilic molecules, which in turn are controlled by the Accessible surface area and the Hydropathy Index of side chain as well as the solubility of amino acid.
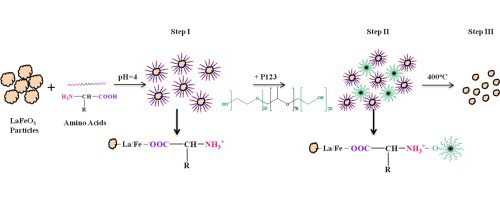
[76] E.D. Koutsouroubi, I.T. Papadas & G.S. Armatas. Ordered Mesoporous Polyoxometalate-Organosilica Frameworks as Efficient Photocatalysts for Hydrogen Evolution Reaction. ChemPlusChem, 81, 947–954 (2016).
Polyoxometalate clusters have been recently established as promising nanomaterials for photocatalytic water splitting. Here, the synthesis is reported of mesoporous polymers composed of a 3D porous network of lacunary [XM11O39]q− (XM11; X=P, Si; M=W, Mo) polyoxometalate units connected by ethano‐bridged silsesquioxane linkers through a block copolymer‐templated crosslinking polymerization of 1,2‐bis(triethoxysilyl)ethane in acidic solution. The resulting materials feature an ordered mesostructured ethane–silica (MES) framework that hosts a high density of accessible polyoxometalate clusters, which allows for efficient catalytic reactions. XM11/MES hybrid polymers have a relatively high activity for the hydrogen evolution reaction with remarkable cycle stability under UV/Vis light irradiation, without the need for co‐catalysts or additional photosensitizers. It is also shown that the photocatalytic efficiency of these materials arises from the nanoscale pore structure, high surface area and chemical manipulation of the electronic band structure of constituting heteropolyoxo clusters.
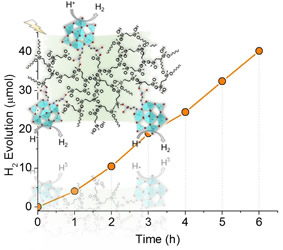
(see our Cover Picture, ChemPlusChem, 81, 1014, 2016)
[75] I. Tamiolakis, I.T. Papadas, K. Spyridopoulos & G.S. Armatas. Mesoporous Assembled Structures of Cu2O and TiO2 Nanoparticles for Highly Efficient Photocatalytic Hydrogen Generation from Water. RSC Adv., 6, 54848–54855 (2016).
Photocatalytic water splitting to produce hydrogen using solar energy is a particularly attractive solution to increasing energy demands. However, to be of practical use, semiconductor electrodes need to be made of inexpensive, abundant elements and have a high, yet stable, photocatalytic H2-production activity. Here we report the first demonstration of 3D mesoporous networks of Cu2O and TiO2 nanoparticles as highly efficient photocatalysts for hydrogen generation from water. These assembled structures feature a highly accessible pore surface that exposes a large fraction of anatase TiO2 and Cu2O nanoparticles to electrolytes, and has a small grain size of the constituent nanocrystals, which lead to excellent activity for H2 evolution via a UV-visible light-driven reduction of protons. Catalytic results associated with optical UV-vis/NIR absorption and photoluminescence (PL) data indicated that the large separation of photogenerated electrons and holes at the Cu2O–TiO2 p–n junctions was the main reason attributed to the improved photochemical performance. Consequently, the mesoporous Cu2O/TiO2 catalyst containing ∼1.5 wt% Cu reaches an average H2 evolution rate of ∼542 μmol h−1 (or ∼36 133 μmol h−1 g−1) with an apparent quantum efficiency (QE) of 13.5% at 365 nm and an incident photon conversion efficiency of ∼4.1% under UV-visible light illumination (360–780 nm), which is one of the best HER activities among TiO2-based semiconductor systems reported to date.
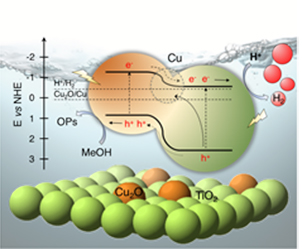
[74] I.T. Papadas, I. Vamvasakis, I. Tamiolakis & G.S Armatas. Templated Self-Assembly of Colloidal Nanocrystals into Three-Dimensional Mesoscopic Structures: A Perspective on the Synthesis and Catalytic Prospects. Chem. Mater., 28, 2886–2896 (2016).
Inorganic nanocrystals (NCs) of uniform size are of eminent importance for the fields of catalysis, energy storage and conversion, and electrochemical devices. Current efforts in science of nanoparticles have focused on the propensity of discrete colloidal NCs to assemble into two- or three-dimensional porous architectures. This aspect becomes important, especially, in applications where mass transport-related effects are a concern, such as in catalysis, separation, and chemical sensing. In this perspective, we describe how colloidal nanocrystals can be used as functional building blocks to construct highly porous networks with large and accessible surface area. The synthesis of these mesostructured assemblies, however, is not a simple process and often requires more sophisticated and elegant processing steps. We focused particularly on the potential of a polymer templating technique for the construction of ordered mesostructured assemblies of metal oxide and metal chalcogenide NCs and concentrate on the application of these materials to catalysis.
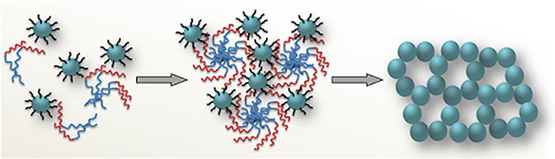
(*Highlighted by J.M. Buriak. Up-and-Coming Perspectives: Share the Excitement of Top Early Career Researchers in Materials Chemistry. Chem. Mater., 28, 4083−4084, 2016)
[73] Ch. Flouraki, M. Kaliva, I.T. Papadas, G.S. Armatas & M. Vamvakaki. Nanoporous Polystyrene-Porphyrin Nanoparticles for Selective Gas Separation. Polym. Chem., 7, 3026–3033 (2016).
Highly cross-linked polystyrene–porphyrin nanoparticles were synthesized by a facile approach using emulsion free-radical copolymerization of styrene (St) with a tetra-functional cross-linker, 5,10,15,20-tetrakis(4-phenylmethacrylate)-21H,23H-porphine (PO), and a bifunctional cross-linker, divinylbenzene (DVB), in aqueous solution. Two samples were prepared, PO-St-DVB(1) and PO-St-DVB(2), with 0.7 and 2.6 mol% PO, respectively. The cross-link density of the particles increased with the increase of the PO content at a constant St/cross-linkers mol feed ratio. The morphology and the size of the polymer particles were studied by scanning and transmission electron microscopies. Gas adsorption measurements showed that the polystyrene–porphyrin nanoparticles possessed an inherently large surface area when dried by supercritical CO2 from their dispersions in EtOH or DMF. In particular, the sample with the highest PO content (PO-St-DVB(2)) exhibited a Brunauer–Emmett–Teller (BET) surface area up to 334 m2 g−1 (282 m2 g−1 calculated from CO2 adsorption) with a total pore volume of 0.37 cm3 g−1 when dried from EtOH. Analysis of the CO2 and CH4 adsorption data using the ideal adsorption solution theory revealed that the PO-St-DVB(2) nanoparticles exhibited an adsorption selectivity for CO2 over CH4 of 25 at 263 K and 12 at 273 K, rendering them attractive candidates for use in CO2/CH4 separation and carbon dioxide sequestration processes.
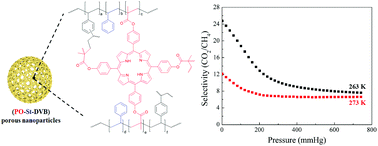
[72] D. Andreou, D. Iordanidou, I. Tamiolakis, G.S. Armatas & I.N. Lykakis. Reduction of Nitroarenes into Aryl Amines and N-Aryl hydroxylamines via Activation of NaBH4 and Amonia-Borane Complexes by Ag/TiO2 Catalyst. Nanomaterials, 6, 54–66 (2016).
In this study, we report the fabrication of mesoporous assemblies of silver and TiO2 nanoparticles (Ag/MTA) and demonstrate their catalytic efficiency for the selective reduction of nitroarenes. The Ag/TiO2 assemblies, which show large surface areas (119–128 m2·g−1) and narrow-sized mesopores (ca. 7.1–7.4 nm), perform as highly active catalysts for the reduction of nitroarenes, giving the corresponding aryl amines and N-aryl hydroxylamines with NaBH4 and ammonia-borane (NH3BH3), respectively, in moderate to high yields, even in large scale reactions (up to 5 mmol). Kinetic studies indicate that nitroarenes substituted with electron-withdrawing groups reduced faster than those with electron-donating groups. The measured positive ρ values from the formal Hammett-type kinetic analysis of X-substituted nitroarenes are consistent with the proposed mechanism that include the formation of possible [Ag]-H hybrid species, which are responsible for the reduction process. Because of the high observed chemo selectivities and the clean reaction processes, the present catalytic systems, i.e., Ag/MTA-NaBH4 and Ag/MTA-NH3BH3, show promise for the efficient synthesis of aryl amines and N-aryl hydroxylamines at industrial levels.
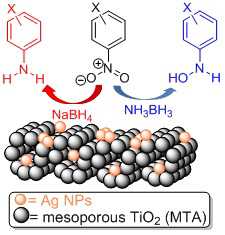
[71] S. Rapti, A. Pournara, D. Sarma, I.T. Papadas, G.S. Armatas, Y.S. Hassan, M.G. Kanatzidis & M.J. Manos. Rapid, Green and Inexpensive Synthesis of High Quality UiO-66 Amino-Functionalized Material and Its Composite with Alginic Acid Showing Great Efficiency for Removal of Hexavalent Chromium from Industrial Waste. Inorg. Chem. Front., 3, 635-644 (2016).
We describe a new synthetic method for the isolation of the UiO-66 amino-functionalized material (called metal organic resin-1, MOR-1) and its composite with alginic acid (HA). MOR-1 can be prepared in high yield (∼70%) and purity within an hour via a reflux reaction of ZrCl4 and 2-amino-terephthalic acid in acifidied aqueous solution, whereas addition of sodium alginate to the fine suspension of MOR-1 resulting from the reflux synthesis affords the MOR-1-HA composite. This inexpensive, green and fast preparation method results in UiO-66 amino-functionalized materials (MOR-1 and MOR-1-HA) of the same quality and microporous features as those of compounds isolated with the slower solvothermal synthesis involving toxic and costly organic solvents. Field Emission-Scanning Electron Microscopy (FE-SEM) studies revealed that MOR-1 consists of spongy nanoparticles (150–300 nm in size), whereas MOR-1-HA nanoparticles are relatively compact. Thus, for the first time we could visualize the effect of alginic acid partially coating the surface of the MOR particles. The composite prepared by this method can be successfully utilized as a stationary phase, mixed with sand, in an anion-exchange column. The column shows excellent hexavalent chromium sorption properties and can be easily regenerated and reused several times with almost no loss of its initial Cr(VI) removal capacity. Remarkably, this ion exchange column is capable of eliminating Cr(VI) ions from chrome plating wastewater samples, thus indicating its potential for applications in industrial wastewater treatment.

(*Highlighted by blogs.rsc.org, 2016)
[70] I.T. Papadas, S. Fountoulaki, I.N. Lykakis & G.S. Armatas. Controllable Synthesis of Mesoporous Iron Oxide Nanoparticle Assemblies for Chemoselective Catalytic Reduction of Nitroarenes. Chem. Eur. J., 22, 4600–4607 (2016).
Iron(III) oxide is a low‐cost material with applications ranging from electronics to magnetism, and catalysis. Recent efforts have targeted new nanostructured forms of Fe2O3 with high surface area‐to‐volume ratio and large pore volume. Herein, the synthesis of 3D mesoporous networks consisting of 4–5 nm γ‐Fe2O3 nanoparticles by a polymer‐assisted aggregating self‐assembly method is reported. Iron oxide assemblies obtained from the hybrid networks after heat treatment have an open‐pore structure with high surface area (up to 167 m2 g−1) and uniform pores (ca. 6.3 nm). The constituent iron oxide nanocrystals can undergo controllable phase transition from γ‐Fe2O3 to α‐Fe2O3 and to Fe3O4 under different annealing conditions while maintaining the 3D structure and open porosity. These new ensemble structures exhibit high catalytic activity and stability for the selective reduction of aryl and alkyl nitro compounds to the corresponding aryl amines and oximes, even in large‐scale synthesis.
[69] S. Fountoulaki, P.L. Gkizis, T.S. Symeonidis, E. Kaminioti, A. Karina, I. Tamiolakis, G.S. Armatas & I.N. Lykakis. Titania-Supported Gold Nanoparticles Catalyze the Selective Oxidation of Amines into Nitroso Compounds in the Presence of Hydrogen Peroxide. Adv. Synth. Catal., 358, 1500–1508 (2016).
In this article, the catalytic activity of titania‐supported gold nanoparticles (Au/TiO2) was studied for the selective oxidation of amines into nitroso compounds using hydrogen peroxide (H2O2). Gold nanoparticles deposited on Degussa P25 polymorphs of titania (TiO2) have been found to promote the selective formation of a variety of nitroso arenes in high yields and selectivities, even in a large‐scale synthesis. In contrast, alkyl amines are oxidized to the corresponding oximes under the examined conditions. Kinetic studies indicated that aryl amines substituted with electron‐donating groups are oxidized faster than the corresponding amines bearing an electron‐withdrawing functionality. A Hammett‐type kinetic analysis of a range of para‐X‐substituted aryl amines implicates an electron transfer (ET) mechanism (ρ=−1.15) for oxidation reactions with concomitant formation of the corresponding N‐aryl hydroxylamine as possible intermediate. We also show that the oxidation protocol of aryl amines in the presence of 1,3‐cyclohexadiene leads in excellent yields to the corresponding hetero Diels–Alder adducts between the diene and the in situ formed nitrosoarenes.
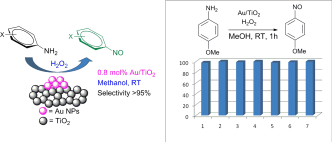
(*Highlighted by Synfacts, 12, 876, 2016; ChemInform, 47, 37, 2016)
[68] S. Rapti, A. Pournara, D. Sarma, I. Papadas, G.S. Armatas, A. Tsipis, T. Lazarides, M.G. Kanatzidis & M. Manos. Selective Capture of Hexavalent Chromium from an Anion-Exchange Column of Metal Organic Resin-Alginic Acid Composite. Chem. Sci., 7, 2427–2436 (2016).
We report an anion exchange composite material based on a protonated amine-functionalized metal–organic framework, denoted Metal Organic Resin-1 (MOR-1), and alginic acid (HA). MOR-1–HA material shows an exceptional capability to rapidly and selectively sorb Cr(VI) under a variety of conditions and in the presence of several competitive ions. The selectivity of MOR-1–HA for Cr(VI) is shown to be the result of strong O3CrVI⋯NH2 interactions. The composite sorbent can be successfully utilized in an ion-exchange column, in contrast to pristine MOR-1 which forms fine suspensions in water passing through the column. Remarkably, an ion exchange column with only 1% wt MOR-1–HA and 99% wt sand (an inert and inexpensive material) is capable of reducing moderate and trace Cr(VI) concentrations to well below the acceptable safety limits for water. The relatively low cost of MOR-1–HA/sand column and its high regeneration capability and reusability make it particularly attractive for application in the remediation of Cr(VI)-bearing industrial waste.
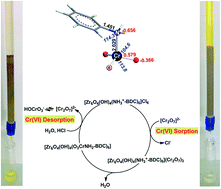
[67] V.I. Markoulaki, I.T. Papadas, I. Kornarakis & G.S. Armatas. Synthesis of Ordered Mesoporous CuO/CeO2 Composite Frameworks as Anode Catalysts for Water Oxidation. Nanomaterials, 5, 1971–1984 (2015).
Cerium-rich metal oxide materials have recently emerged as promising candidates for the photocatalytic oxygen evolution reaction (OER). In this article, we report the synthesis of ordered mesoporous CuO/CeO2 composite frameworks with different contents of copper(II) oxide and demonstrate their activity for photocatalytic O2 production via UV-Vis light-driven oxidation of water. Mesoporous CuO/CeO2 materials have been successfully prepared by a nanocasting route, using mesoporous silica as a rigid template. X-ray diffraction, electron transmission microscopy and N2 porosimetry characterization of the as-prepared products reveal a mesoporous structure composed of parallel arranged nanorods, with a large surface area and a narrow pore size distribution. The molecular structure and optical properties of the composite materials were investigated with Raman and UV-Vis/NIR diffuse reflectance spectroscopy. Catalytic results indicated that incorporation of CuO clusters in the CeO2 lattice improved the photochemical properties. As a result, the CuO/CeO2 composite catalyst containing ~38 wt % CuO reaches a high O2 evolution rate of ~19.6 µmol·h−1 (or 392 µmol·h−1·g−1) with an apparent quantum efficiency of 17.6% at λ = 365 ± 10 nm. This OER activity compares favorably with that obtained from the non-porous CuO/CeO2 counterpart (~1.3 µmol·h−1) and pure mesoporous CeO2 (~1 µmol·h−1).
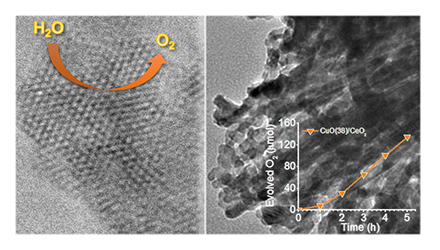
(*This article belongs to the Special Issue "Frontiers in Mesoporous Nanomaterials", Nanomaterials, 6, 15, 2016)
[66] K.S. Subrahmanyam, C.D. Malliakas, D. Sarma, G.S. Armatas, J. Wu & M.G. Kanatzidis. Ion-exchangeable Molybdenum-sulfide Porous Chalcogel: Gas Adsorption and Capture of Iodine and Mercury. J. Am. Chem. Soc., 137, 13943–13948 (2015).
We report the synthesis of ion-exchangeable molybdenum sulfide chalcogel through an oxidative coupling process, using (NH4)2MoS4 and iodine. After supercritical drying, the MoSx amorphous aerogel shows a large surface area up to 370 m2/g with a broad range of pore sizes. X-ray photoelectron spectroscopic and pair distribution function analyses reveal that Mo6+ species undergo reduction during network assembly to produce Mo4+-containing species where the chalcogel network consists of [Mo3S13] building blocks comprising triangular Mo metal clusters and S22– units. The optical band gap of the brown-black chalcogel is ∼1.36 eV. The ammonium sites present in the molybdenum sulfide chalcogel network are ion-exchangeable with K+ and Cs+ ions. The molybdenum sulfide aerogel exhibits high adsorption selectivities for CO2 and C2H6 over H2 and CH4. The aerogel also possesses high affinity for iodine and mercury.
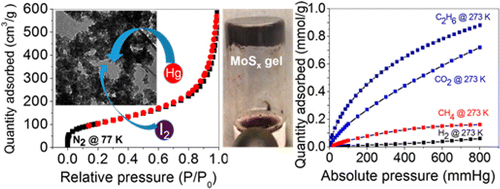
[65] A. Douvali, G.S. Papaefstathiou, M.P. Gullo, A. Barbieri, A.C. Tsipis, C.D. Malliakas, M.G. Kanatzidis, I. Papadas, G.S. Armatas, A.G. Hatzidimitriou, T. Lazarides & M.J. Manos. Alkaline Earth Metal Ion/Dihydroxy–Terephthalate MOFs: Structural Diversity and Unusual Luminescent Properties. Inorg. Chem., 54, 5813–5826 (2015).
Alkaline earth (group 2) metal ion organic frameworks (AEMOFs) represent an important subcategory of MOFs with interesting structures and physical properties. Five MOFs, namely, [Mg2(H2dhtp)2(μ-H2O)(NMP)4] (AEMOF-2), [Mg2(H2dhtp)1.5(DMAc)4]Cl·DMAc (AEMOF-3), [Ca(H2dhtp)(DMAc)2] (AEMOF-4), [Sr3(H2dhtp)3(DMAc)6]·H2O (AEMOF-5), and [Ba(H2dhtp)(DMAc)] (AEMOF-6) (H4dhtp = 2,5-dihydroxy-terepthalic acid; DMAc = N,N-dimethylacetamide; NMP = N-methylpyrrolidone), are presented herein. The reported MOFs display structural variety with diverse topologies and new structural features. Interestingly, AEMOF-6 is the first example of a Ba2+–H2dhtp2– MOF, and AEMOF-5 is only the second known Sr2+–H2dhtp2– MOF. Detailed photoluminescence studies revealed alkaline earth metal ion-dependent fluorescence properties of the materials, with the heavier alkaline earth metal ions exhibiting red-shifted emission with respect to the lighter ions at room temperature. A bathochromic shift of the emission was observed for the MOFs (mostly for AEMOF-3 and AEMOF-4) at 77 K as a result of excited state proton transfer (ESIPT), which involves an intramolecular proton transfer from a hydroxyl to an adjacent carboxylic group of the H2dhtp2– ligand. Remarkably, AEMOF-6 displays rare yellow fluorescence at room temperature, which is attractive for solid state lighting applications. To probe whether the alkaline earth metal ions are responsible for the unusual luminescence properties of the reported MOFs, the potential energy surfaces (PESs) of the ground, S0, and lowest energy excited singlet, S1, states of model complexes along the intramolecular proton transfer coordinate were calculated by DFT and TD-DFT methods.
[64] I. Vamvasakis, K.S. Subrahmanyam, M.G. Kanatzidis & G.S Armatas. Template-Directed Assembly of Metal-Chalcogenide Nanocrystals into Ordered Mesoporous Networks. ACS Nano, 9, 4419–4426 (2015).
Although great progress in the synthesis of porous networks of metal and metal oxide nanoparticles with highly accessible pore surface and ordered mesoscale pores has been achieved, synthesis of assembled 3D mesostructures of metal–chalcogenide nanocrystals is still challenging. In this work we demonstrate that ordered mesoporous networks, which comprise well-defined interconnected metal sulfide nanocrystals, can be prepared through a polymer-templated oxidative polymerization process. The resulting self-assembled mesostructures that were obtained after solvent extraction of the polymer template impart the unique combination of light-emitting metal chalcogenide nanocrystals, three-dimensional open-pore structure, high surface area, and uniform pores. We show that the pore surface of these materials is active and accessible to incoming molecules, exhibiting high photocatalytic activity and stability, for instance, in oxidation of 1-phenylethanol into acetophenone. We demonstrate through appropriate selection of the synthetic components that this method is general to prepare ordered mesoporous materials from metal chalcogenide nanocrystals with various sizes and compositions.
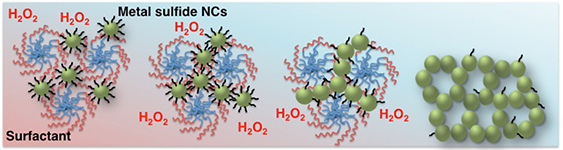
[63] I.T. Papadas, K.S. Subrahmanyam, M.G. Kanatzidis & G.S Armatas. Templated Assembly of BiFeO3 Nanocrystals into 3D Mesoporous Networks for Catalytic Applications. Nanoscale, 7, 5737–5743 (2015).
The self-assembly of uniform nanocrystals into large porous architectures is currently of immense interest for nanochemistry and nanotechnology. These materials combine the respective advantages of discrete nanoparticles and mesoporous structures. In this article, we demonstrate a facile nanoparticle templating process to synthesize a three-dimensional mesoporous BiFeO3 material. This approach involves the polymer-assisted aggregating assembly of 3-aminopropanoic acid-stabilized bismuth ferrite (BiFeO3) nanocrystals followed by thermal decomposition of the surfactant. The resulting material consists of a network of tightly connected BiFeO3 nanoparticles (∼6–7 nm in diameter) and has a moderately high surface area (62 m2 g−1) and uniform pores (ca. 6.3 nm). As a result of the unique mesostructure, the porous assemblies of BiFeO3 nanoparticles show an excellent catalytic activity and chemical stability for the reduction of p-nitrophenol to p-aminophenol with NaBH4.

[62] E.D. Koutsouroubi, A.K. Xylouri & G.S. Armatas. Mesoporous Polyoxometalate Cluster–Crosslinked Organosilica Frameworks Delivering Exceptionally High Photocatalytic Activity. Chem. Commun., 51, 4481–4484 (2015).
Mesoporous framework materials comprising lacunary [SiW11O39]8− polyoxometalate clusters covalently connected by ethane-bridged silsesquioxane linkers were synthesized through a block copolymer-templated cross-linking polymerization of 1,2-bis(triethoxysilyl)ethane in acid solution. These new hybrid materials, which exhibit a high density of catalytic sites, large pore surface and ordered pore structure, are shown to be highly effective in the photocatalytic oxidation of aryl alcohols with molecular oxygen.
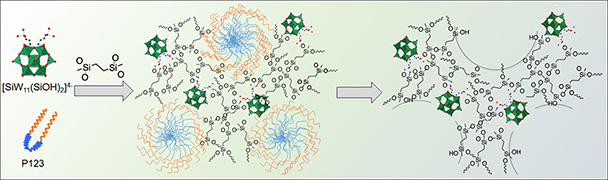
[61] A. Douvali, A.C. Tsipis, S.V. Eliseeva, S. Petoud, G.S. Papaefstathiou, C.D. Malliakas, I. Papadas, G.S. Armatas, M.G. Kanatzidis, T. Lazarides & M.J. Manos. Turn-On Luminescence Sensing and Real Time Detection of Traces of Water in Organic Solvents by a Flexible Metal Organic Framework. Angew. Chem. Int. Ed., 54, 1651–1656 (2015).
The development of efficient sensors for the determination of the water content in organic solvents is highly desirable for a number of chemical industries. Presented herein is a Mg2+ metal–organic framework (MOF), which exhibits the remarkable capability to rapidly detect traces of water (0.05–5 % v/v) in various organic solvents through an unusual turn‐on luminescence sensing mechanism. The extraordinary sensitivity and fast response of this MOF for water, and its reusability make it one of the most powerful water sensors known.
[60] I. Papadas, J.A. Christodoulides, G. Kioseoglou & G.S. Armatas. High Surface Area Ordered Mesoporous BiFeO3 Semiconductor with Efficient Water Oxidation Activity. J. Mater. Chem. A, 3, 1587–1593 (2015).
Bismuth ferrite (BiFeO3) is an important multiferroic oxide material because of its unique magnetic and ferroelectric properties. Here, we synthesize for the first time a highly ordered mesoporous BiFeO3 semiconductor using tartaric acid-assisted growth of the BiFeO3 compound inside the pores of a carbon template. Powder X-ray diffraction (XRD), transmission electron microscopy (TEM) and N2 physisorption measurements reveal that the template-free material possesses a three-dimensional hexagonal mesostructure with a large internal BET surface area (141 m2 g−1) and narrow sized pores (ca. 4 nm). Also, the pore walls comprise single-phase BiFeO3 nanocrystals according to the high-resolution TEM, electron diffraction and magnetic experiments. The mesoporous BiFeO3 shows high activity for the photocatalytic oxygen evolution reaction (OER) under UV-visible light, affording an average oxygen evolution rate of 66 μmol h−1 g−1. We also show that the propensity of photogenerated holes for the OER can be significantly enhanced when 1 wt% Au nanoparticles are deposited on the BiFeO3 surface. The Au/BiFeO3 heterostructure exerts excellent OER activity (586 μmol h−1 g−1) and long-term cycling stability, raising the possibility for the design of effective and robust OER photocatalysts.
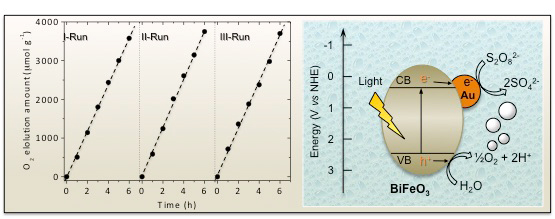
[59] I. Lykakis, T.S. Symeonidis, I. Tamiolakis & G.S. Armatas. Green photocatalytic organic transformations by polyoxometalates vs mesoporous TiO2 nanoparticles: Selective aerobic oxidation of alcohols. Photochem. Photobiol. Sci., 114, 563–568 (2015).
In this study, the catalytic activity of decatungstate (W10O324−) supported on mesoporous TiO2 nanoparticle assemblies (DT-MTA) was compared with that of homogeneous [Bu4N]4W10O32 catalysts under mild conditions. Our experiments showed that both catalytic systems achieve exceptionally high activity and selectivity under UV-visible light oxidation of various para-substituted aryl alcohols, using molecular oxygen as a “green” oxidant. The chemoselective transformation of aryl alcohols into the corresponding ketones was investigated with gas chromatography (GC) and NMR spectroscopy. Product analysis and kinetic results also indicated that these photooxidation reactions proceed via both electron transfer (ET) and hydrogen atom transfer (HAT) mechanisms over the DT-MTA catalyst, with the former one as the predominant, whereas a HAT route was adopted to account for the decatungstate homogeneous catalyzed reactions.
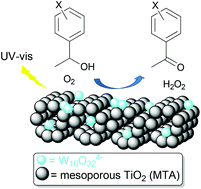
[58] I. Tamiolakis, I.N. Lykakis & G.S. Armatas. Mesoporous CdS-sensitized TiO2 nanoparticle assemblies with enhanced photocatalytic properties: Selective aerobic oxidation of benzyl alcohols. Catal. Today, 250, 180–186 (2015).
Mesoporous TiO2-based semiconductors with visible-light response are promising materials for photocatalytic and photoelectrochemistry applications. In this work, we have used surfactant-assisted aggregating assembly of CdS and TiO2 nanocrystals to assemble mesoporous binary CdS-TiO2 heterostructure. The product features a three-dimensional network of interconnected CdS quantum dots and anatase TiO2 nanoparticles and exhibits large internal BET surface area (157 m2 g−1) and uniform pores (ca. 7.5 nm). Catalytic experiments showed an exceptionally high catalytic activity of these mesophases under UV–visible light oxidation of various para-substituted aryl alcohols, using molecular oxygen as oxidant. Moreover, product analysis and kinetic results indicated that these photooxidation reactions proceed via an electron transfer route from alcohol substrate to the excited states of the catalyst.
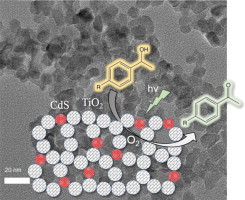
[57] I. Papadas, S. Fountoulaki, I.N. Lykakis & G.S. Armatas. Mesoporous Au-loaded Fe2O3 Nanoparticle Assemblies for Chemoselective Reduction of Nitroarenes. Mater. Res. Soc. Symp. Proc., 1749, 1–6 (2014).
In this article, we report the synthesis of unique mesoporous Au-loaded Fe2O3nanoparticle assemblies (Au/Fe2O3-NPAs) through a surfactant-assisted aggregating assembly method. The resulting network structure, which composed of small Au nanocrystals (ca. 5 nm) finely dispersed on surface of Fe2O3 NPs (ca. 6–7 nm), possesses a 3D open-pore structure with a BET surface area of 123 m2g-1 and uniform mesopores (∼4.5 nm). Au/Fe2O3-NPAs showed high catalytic activity and chemical stability for the selective transformation of nitroaromatic compounds into the corresponding amines, using 1,1,3,3-tetramethyl disiloxane as reducing agent at ambient conditions.

[56] P.L. Gkizis, I. Kalara-Lafkioti, D. Varelas, I. Tamiolakis, G.S. Armatas & I.N. Lykakis. Efficient and selective oxidation of aromatic amines into nitrosoarenes catalyzed by supported polyoxometalates. Biointerface Res. Appl. Chem., 4, 857–860 (2014).
In this article we studied the selective oxidation of various aryl amines into the corresponding nitrosoarenes, over polyoxometalates anions supported on mesoporous TiO2 nanoparticles assemblies (POM-MTA) using H2O2 as “green” oxidant. The use of supported catalyst for heterogeneous catalysis offers several advantages on the catalyst reusability and the regio- and chemo-selectivity of the reaction process. Our catalytic experiments showed an exceptionally high activity of these catalysts towards aryl amines oxidation in the presence of H2O2, with high yields and selectivity at ambient conditions and short reaction times.
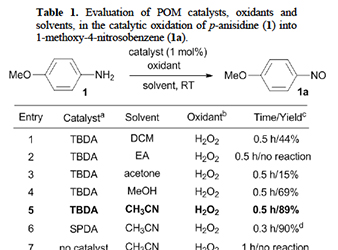
[55] E. Skliri, I. Lykakis & G.S. Armatas. Ordered Mesoporous V2O5-WO3 Composite Catalysts for Efficient Oxidation of Aryl Alcohol. RSC Adv., 4, 46170–46178 (2014).
Multicomponent mesoporous metal oxides show promise in the area of heterogeneous catalysis due to the synergetic interactions between the framework components and the high internal surface area. In this study, we present the synthesis of ordered mesoporous tungsten(VI) oxide–vanadium oxide (V2O5) nanocomposite frameworks via a two-step wet chemical deposition and nanocasting process and demonstrate that they exhibit high catalytic activity and stability for the oxidation of aryl alcohols, using tert-butyl hydroperoxide (t-BuOOH) as oxidant. X-ray diffraction, transmission electron microscopy and nitrogen porosimetry results indicate that the template-free materials possess a 3D mesoscopic structure of discernible domains of parallel-arranged nanorods and have an internal pore surface with narrow mesopores. The chemical composition and molecular structure of the mesoporous matrix were determined with elemental X-ray microanalysis (EDS), diffuse reflectance ultraviolet-visible (UV-vis) and Raman spectroscopy. Our catalytic results indicate that a small addition of V2O5 into the lattice of WO3 has a beneficial effect on the catalytic performance. Thus, the 4% V2O5-loaded WO3 catalyst shows a large improvement in the oxidation of various para-substituted aryl alcohols with respect to the pure mesoporous WO3, giving good-to-high yields (ca. 80–100%) of the target products within 1–4 h reaction time.
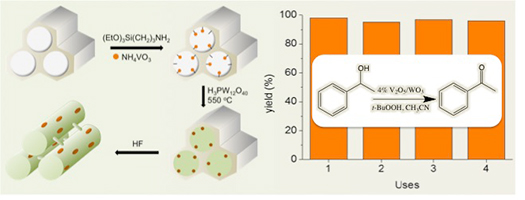
[54] S. Fountoulaki, V. Daikopoulou, P. Gkizis, I. Tamiolakis, G.S. Armatas & I.N. Lykakis. Mechanistic studies in the reduction of nitro arenes by NaBH4 or hydrosilanes catalyzed by supported gold nanoparticles. ACS Catal., 4, 3504–3511 (2014).
Herein, we show that mesoporous titania-supported gold nanoparticle assemblies (Au/MTA) catalyze the activation of NaBH4 and 1,1,3,3-tetramethyl disiloxane (TMDS) compounds, which act as transfer hydrogenation agents for the reduction of nitroarenes to the corresponding anilines in moderate to high yields. On the other hand, nitroalkanes are reduced to the corresponding diazo and hydrazo compounds under the studied conditions. The substantial measured primary kinetic isotope effects found here suggested that B–H bond cleavage occurs in a rate-determining step and [Au]–H active hybrids are formed, which are responsible for the reduction of nitroarenes to the corresponding amines. Formal Hammett-type kinetic analysis of a range of para-X-substituted nitroarenes lends support to this hypothesis. Nitro compounds substituted with electron-withdrawing groups were reduced faster than the corresponding compounds with electron-donating groups. The presence of water enhanced the catalytic activity of Au/MTA in aprotic solvents. Nuclear magnetic resonance studies support the formation of the corresponding hydroxylamines as the only intermediate products. On the basis of the high observed chemoselectivities and the fast and clean reaction processes, these catalytic systems, i.e., Au/MTA-NaBH4 and Au/MTA-TMDS, show promise for the efficient synthesis of aromatic amines at industrial levels.
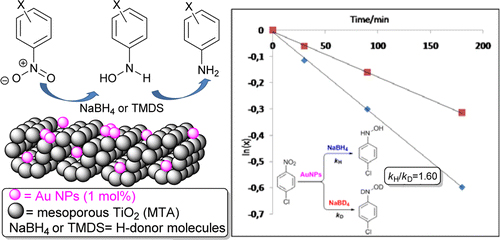
[53] I. Kornarakis, I.N. Lykakis, N. Vordos & G.S. Armatas. Efficient Visible-Light Photocatalytic Activity by Band Alignment in Mesoporous Ternary Polyoxometalate/Ag2S/CdS Semiconductors. Nanoscale, 6, 8694–8703 (2014).
Porous multicomponent semiconductor materials show improved photocatalytic performance due to the large and accessible pore surface area and high charge separation efficiency. Here we report the synthesis of well-ordered porous polyoxometalate (POM)–Ag2S–CdS hybrid mesostructures featuring a controllable composition and high photocatalytic activity via a two-step hard-templating and topotactic ion-exchange chemical process. Ag2S compounds and polyoxometalate cluster anions with different reduction potentials, such as PW12O403−, SiW12O404− and PMo12O403−, were employed as electron acceptors in these ternary heterojunction photocatalysts. Characterization by small-angle X-ray scattering, X-ray diffraction, transmission electron microscopy and N2 physisorption measurements showed hexagonal arrays of POM–Ag2S–CdS hybrid nanorods with large internal BET surface areas and uniform mesopores. The Keggin structure of the incorporated POM clusters was also verified by elemental X-ray spectroscopy microanalysis, infrared and diffuse-reflectance ultraviolet-visible spectroscopy. These new porous materials were implemented as visible-light-driven photocatalysts, displaying exceptional high activity in aerobic oxidation of various para-substituted benzyl alcohols to the corresponding carbonyl compounds. Our experiments show that the spatial separation of photogenerated electrons and holes at CdS through the potential gradient along the CdS–Ag2S–POM interfaces is responsible for the increased photocatalytic activity.
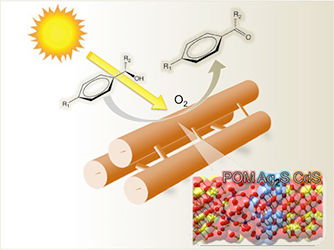
[52] I. Tamiolakis, I.N. Lykakis & G.S. Armatas. Mesoporous Au–TiO2 Nanoparticle Assemblies as Efficient Catalysts for the Chemoselective Reduction of Nitro Compounds. Mater. Res. Soc. Symp. Proc., 1641, 1–6 (2014).
Here, we propose novel mesoporous Au-loaded TiO2 nanoparticle assemblies (Au-MTA) as highly effective catalysts for the reduction of nitroaromatic compounds into the corresponding aryl amine products. The obtained materials possess a continuous network of interconnected gold and anatase TiO2 (ca. 9 nm in size) nanoparticles with controllable gold particle size (i.e. ranging from ∼3.2 to ∼9.4 nm) and exhibit large and accessible pore surface area (ca. 100–160 m2/g), as evidenced by SAXS, XRD, TEM and N2 physisorption measurements. Interestingly, the Au-MTA mesophases have exhibited remarkable activity and selectivity for the reduction of nitro into amine groups using NaBH4 as reducing agent. Indeed, the Au loading and particle size have a key effect on hydrogenation reactions, affecting significantly the yield and product composition.
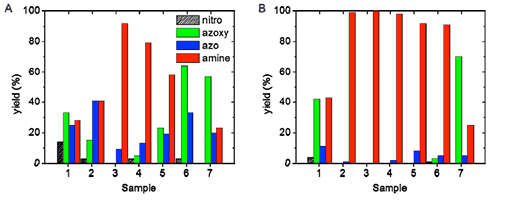
[51] M. Manos, G.S. Papaefstathiou, K.S. Subrahmanyam, C.D. Malliakas, G.S. Armatas & M.G. Kanatzidis. A unique microporous copper-trimesate selenite with high selectivity for CO2. CrystEngComm, 16, 3483–3486 (2014).
A copper trimesate selenite compound is reported which shows a robust 3-D porous structure with unprecedented structural characteristics and represents a unique example of a metal inorganic–organic framework with considerable permanent microporosity and selective CO2 sorption properties.
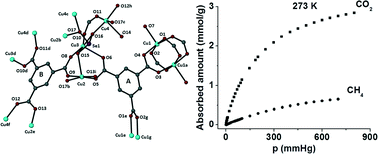
[50] E. Skliri, I.N. Lykakis & G.S. Armatas. Heteropolytungstic acids incorporated in an ordered mesoporous zirconia framework as efficient oxidation catalysts. RSC Adv., 4, 8402–8409 (2014).
Ordered mesoporous composite catalysts consisting of nanocrystalline tetragonal ZrO2 and heteropolytungstic clusters, i.e. 12-phosphotungstic (PTA) and 12-silicotungstic (STA) acids, were prepared via a surfactant-assisted co-polymerization route. According to the X-ray diffraction, transmission electron microscopy and N2 physisorption measurements, the resultant materials possess a well-defined mesoscopic order ranging from wormhole to hexagonal pore structure and exhibit large internal surface area (126–229 m2 g−1) and quite narrow pore size distribution (ca. 2.2–2.6 nm in diameter). Energy dispersive X-ray microanalysis and infrared spectroscopy confirms that the heteropoly clusters are well dispersed within the zirconia matrix, while preserving intact their Keggin structure. The inclusion of PTA and STA clusters in the mesoporous framework has a beneficial effect on the catalytic activity of these materials. Although zirconium oxide and heteropoly acids alone show little catalytic activity, the ZrO2–PTA and ZrO2–STA heterostructures exhibit surprisingly high activity in hydrogen peroxide mediated oxidation of 1,1-diphenyl-2-methylpropene under mild conditions. Indeed, the mesoporous ZrO2–STA composite sample loaded with 5 wt% STA shows a conversion rate that is 17 times higher than the mesoporous ZrO2. The catalytic activity of these materials is related to the spatial distribution of heteropoly acids in zirconia matrix and possible synergistic interactions between the incorporated Keggin units and Zr(IV) oxohydroxide species.
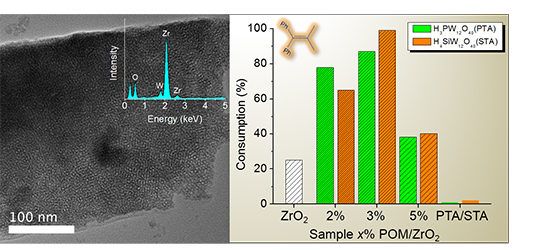
[49] I. Tamiolakis, S. Fountoulaki, N. Vordos, I.N. Lykakis & G.S. Armatas. Mesoporous Au–TiO2 nanoparticle assemblies as efficient catalysts for the chemoselective reduction of nitro compounds. J. Mater. Chem. A, 1, 14311–14319 (2013).
In this article, we demonstrate novel mesoporous Au-loaded TiO2 nanoparticle assemblies (Au–MTA) as high-effective catalysts for the selective transformation of nitroaromatics into the corresponding aryl amine products. These materials feature a three-dimensional open porous structure consisting of interconnected uniform gold and TiO2 nanoparticles, large internal surface area (ca. 104–120 m2 g−1), and narrow mesopores (ca. 7.3–7.6 nm). Au–MTA displays outstanding performance for the selective reduction of nitro into amine groups using sodium borohydride as a reducing agent under ambient conditions. We show that both chemoselectivity and hydrogenation activity of the Au–MTA catalysts are highly related to the Au loading and particle size. As a result the 2% Au–MTA associated with 5 nm sized Au particles was found to be a prominent catalyst for hydrogenation reactions, providing exceptionally high selectivity (96%) and conversion yield (92%) to the corresponding amines. The implication of these results is that mesoporous ensembles of gold and TiO2 nanoparticles should be considered for the synthesis of fine amine chemicals.
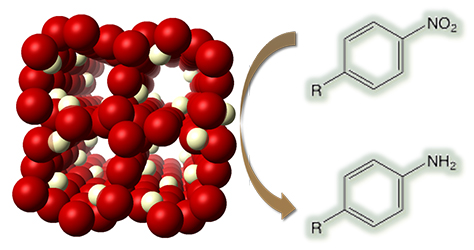
[48] I. Tamiolakis, I.N. Lykakis & G.S. Armatas. Synthesis and Photocatalytic Properties of High-Surface-Area Mesoporous TiO2 Nanoparticle Assemblies. Mater. Res. Soc. Symp. Proc., 1494, 1–6 (2013).
Mesoporous TiO2 nanoparticle assemblies have been synthesized via a surfactant-assisted aggregating process. The products feature a three-dimensional network of interconnected anatase-TiO2 NPs with large internal BET surface area (ca. 142–152 m2g-1) and uniform pores (ca. 7–8 nm). Preliminary catalytic experiments indicated that these mesophases exhibit excellent catalytic activity in UV-visible light oxidation of 1-phenylethanol with molecular oxygen.
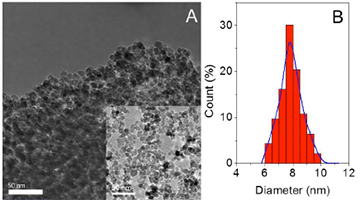
[47] G.S. Armatas. Heterogeneous Polyoxometalate-Containing Mesoporous Catalysts. S.L. Suib (Ed.) in “New and Future Developments in Catalysis: Hybrid Materials, Composites, and Organocatalysts”, Elsevier, Amsterdam (2012).
Mesoporous metal oxides that contain polyoxometalate (POM) compounds are highly attractive materials for a wide range of catalytic applications. The incorporation of POM components within a porous inorganic support may endow intriguing functionalities to the parent materials such as high catalytic activity and recyclability. A substantial progress has been made in the synthesis of new mesoporous polyoxometalate-based catalysts by employing post-synthesis grafting or wet impregnation of POM units onto inorganic supports or direct sol-gel reaction of metal oxide species and POM compounds. This chapter highlights the recent achievements in this area emphasizing the preparation and catalytic properties of these materials.

[46] I. Kornarakis, G. Sopasis, C.J. Milios & G.S. Armatas. Incorporation of a High-spin Heptanuclear [CuII6Gd] Cluster into Carboxyl–functionalized Mesoporous Silica. RSC Adv., 2, 9809–9815 (2012).
We report the immobilization of heptanuclear [Cu6Gd] high-spin magnetic clusters, namely [Cu6Gd(aib)6(OAc)3(NO3)3(OH)3] (where, aib = (CH3)2C(NH2)COO− and OAc = CH3COO−), inside the channels of –COOH-functionalized mesoporous SBA-15 silica. X-ray diffraction, TEM and N2 physisorption experiments show that the well-ordered hexagonal structure of the CA/SBA-15 hybrid material is well preserved and that the [Cu6Gd] clusters are located inside the mesopores. The molecular structure of the incorporated [Cu6Gd] was confirmed by elemental X-ray microanalysis, IR and UV-vis-NIR spectroscopy. Magnetic measurements indicate that the polynuclear complexes retain their magnetic properties during the impregnation process. The host–guest nanocomposite displays strong ferromagnetic interactions that result in a high-spin S = 13/2 ground state.
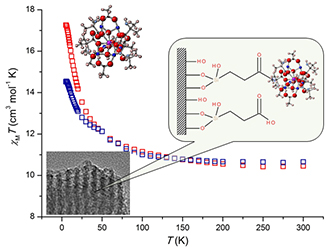
[45] G.G. Papagianni, D.V. Stergiou, G.S. Armatas, M.G. Kanatzidis & M.I. Prodromidis. Synthesis, Characterization and Performance of Polyaniline - Polyoxometalates (XM12, X= P, Si and M= Mo, W) Composites as Electrocatalysts of Bromates. Sensors & Actuators: B. Chem., 173, 346–353 (2012).
Composite materials of the type polyaniline (PANI)–polyoxometalates (POM = XM12, X = P, Si and M = Mo, W) were synthesized and characterized by means of FT-IR, UV–vis, SEM, XRD, SAXS analysis and cyclic voltammetry. Suspensions of 2.5 mg mL−1 PANI–PMo12 in 2% (w/v) poly(ethyleneimine) (PEI) in methanol were prepared and deposited onto graphene oxide-modified graphite electrodes. These were tested as sensors for the ability to electrocatalyze the reduction of bromates by using cyclic voltammetry. Various experimental variables such as the loading of the composite material, the concentration of PEI, the pH and the composition of electrolyte, and the electrode material were optimized. By performing DC amperometry at a fixed potential of −0.3 V, versus a Ag/AgCl reference electrode, in 0.05 M HCl, a linear calibration curve over the concentration range 7.5 × 10−6 to 5.0 × 10−4 M BrO3− was constructed. The 3σ limit of detection and the relative standard deviation of the method were 3.0 × 10−6 M BrO3− and 3% (n = 7, 1 × 10−4 M BrO3−), respectively. The accuracy of the method was established by recovery studies in spiked drinking water and flour extract samples. Recovery was 95.4–100.2%. Finally, the sensors exhibited a remarkable working and storage stability when they kept dry in ambient conditions.

[44] I. Tamiolakis, I.N. Lykakis, A.P. Katsoulidis & G.S. Armatas. One-Pot Synthesis of Highly Crystalline Mesoporous TiO2 Nanoparticle Assemblies with Enhanced Photo-catalytic Activity. Chem. Commun., 48, 6687–6689 (2012).
We report an unprecedented formation of mesoporous titania via a surfactant-assisted aggregating assembly of TiO2 nanoparticles. These mesostructures possess a highly crystalline anatase phase, large and accessible pore surface area, and exhibit superior photocatalytic performance.
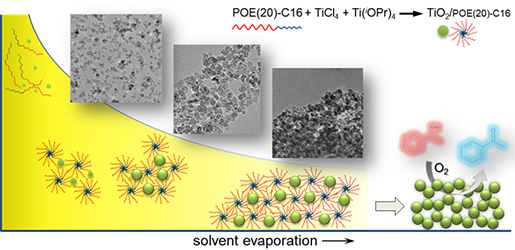
[43] P.J. Pomonis & G.S. Armatas. The Pore Anisotropy in Porous Solids. H.G. Lemu (Ed.) in “Anisotropy Research: New Developments”, Nova Science Publishers, New York, Chap. 3 (2012).
Anisotropy is a term used to indicate the directional dependence of a biological, chemical, geometrical, mechanical or physical property of a system that develops in different ways along different spatial axes. This material behavior is observed in many engineering disciplines, computer graphics, geology and geophysics, medicine, neurosciences and natural sciences. Thus the research approaches within those disciplines accordingly varies. This book has collected contributions of new research developments and the corresponding research results from different disciplines with particular focus on experimental, numerical and theoretical studies of material property variations. Thus, the materials in this book indicate significant directions on new research progresses and are expected to give future research perspectives in anisotropy behavior.

[42] I. Tamiolakis, I.N. Lykakis, A.P. Katsoulidis, C.D. Malliakas & G.S. Armatas. Ordered Mesoporous Cr2O3 Frameworks Incorporating Keggin-type 12-Phosphotungstic Acids as Efficient Catalysts for Oxidation of Benzyl Alcohols. J. Mater. Chem. 22, 6919–6927 (2012).
Ordered mesoporous chromium(III) oxide–phosphotungstic acid (PWA) nanocomposite structures with controllable composition (∼17 to 49 wt% in PWA) have been successfully prepared via an ultrasound-assisted nanocasting route, using mesoporous SBA-15 silica as a rigid mold. These materials possess 3D hexagonal mesostructure, large internal BET surface areas of ∼67 to 80 m2 g−1, and uniform pores of ∼3 to 4 nm size according to small-angle X-ray scattering, high resolution transmission electron microscopy and N2 physisorption. The Keggin-type structure of [PW12O40]3− anions is preserved intact into the Cr2O3 framework, as confirmed by total X-ray diffuse scattering and pair distribution function analysis and infrared and diffuse reflectance ultraviolet-visible (UV-vis) spectroscopy. The integration of regular porosity, large internal surface area, and Cr2O3–PWA composition makes these materials highly promising for applications in oxidation catalysis. Although pure mesoporous Cr2O3 and PWA compounds exhibit low catalytic activity, the mesoporous Cr2O3–PWA composites showed superior activity and selectivity for the oxidation of selected secondary benzyl alcohols, giving good-to-high yields within a short reaction time. Furthermore, the Cr2O3–PWA composite frameworks demonstrated remarkable durability and reusability upon multiple usages without leaching or decomposition of the incorporated PWA clusters. This enhancement is attributed to the synergistic interactions between the PWA and Cr2O3 components as well as the well-ordered open-pore structure and large catalytically active surface area.
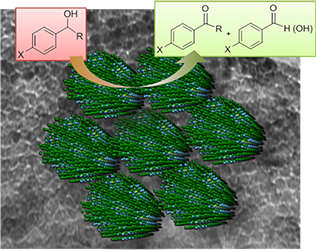
[41] M. Kaliva, G.S Armatas & M. Vamvakaki. Microporous Polystyrene Particles for Selective Carbon Dioxide Capture. Langmuir, 28, 2690–2695 (2012).
This study presents the synthesis of microporous polystyrene particles and the potential use of these materials in CO2 capture for biogas purification. Highly cross-linked polystyrene particles are synthesized by the emulsion copolymerization of styrene (St) and divinylbenzene (DVB) in water. The cross-link density of the polymer is varied by altering the St/DVB molar ratio. The size and the morphology of the particles are characterized by scanning and transmission electron microscopy. Following supercritical point drying with carbon dioxide or lyophilization from benzene, the polystyrene nanoparticles exhibit a significant surface area and permanent microporosity. The dried particles comprising 35 mol % St and 65 mol % DVB possess the largest surface area, ∼205 m2/g measured by Brunauer–Emmett–Teller and ∼185 m2/g measured by the Dubinin–Radushkevich method, and a total pore volume of 1.10 cm3/g. Low pressure measurements suggest that the microporous polystyrene particles exhibit a good separation performance of CO2 over CH4, with separation factors in the range of ∼7–13 (268 K, CO2/CH4 = 5/95 gas mixture), which renders them attractive candidates for use in gas separation processes.
[40] I. Tamiolakis, I.N. Lykakis, A.P. Katsoulidis, M. Stratakis & G.S. Armatas. Mesoporous Cr2O3-Phosphomolybdic Acid Solid Solution Frameworks with High Catalytic Activity. Chem. Mater. 23, 4204–4211 (2011).
Mesoporous nanocomposite frameworks of chromium(III) oxide and 12-phosphomolybdic acid (PMA) compounds were prepared via a “nanocasting” method, using mesoporous silica SBA-15 as the template. Small-angle X-ray scattering, transmission electron microscopy, and nitrogen physisorption characterizations indicate that these materials possess a well-ordered hexagonal mesostructure with high content of PMA clusters (up to 63 wt %) and large internal surface area (up to 165 m2 g–1). The Keggin clusters are incorporated intact into the mesoporous walls forming a Cr2O3–PMA solid-solution structure, according to the X-ray diffraction and X-ray photoelectron and ultraviolet–visible light (UV/vis) spectroscopy. These Cr2O3–PMA nanocomposites demonstrated strong potential for green oxidative processes, exhibiting high catalytic activity and durability in oxidation of 1-phenylethanol using H2O2 as an oxidant.
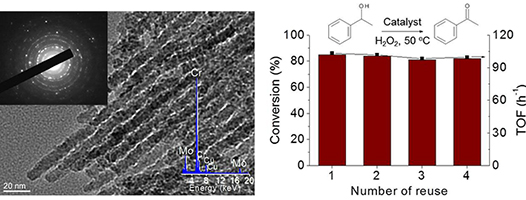
[39] M. Orfanoudaki, I. Tamiolakis, M. Siczek, T. Lis, G.S. Armatas, S.A. Pergantis & C.J. Milios. Unique trigonal prism encapsulated Ln complexes: A [CoII6Eu] and a [CoII6Dy] cage. Dalton Trans., 40, 4793 (2011).
The use of 2-amino-isobutyric acid in Co/Ln chemistry has led to the isolation of two unique [CoII6LnIII] 3d-4f metallic cages in which the LnIII centre (Ln = Eu, Dy) is encapsulated within a CoII6 trigonal prism.
[38] G.S. Armatas, I. Tamiolakis & D.E. Petrakis. Periodically Ordered Mesoporous Co3O4/Heteropoly Acid Composite Frameworks for Catalytic Applications. Mater. Res. Soc. Symp. Proc. 1309, 1–7 (2011).
Mesoporous cobalt oxide-tungstophosphoric acid composite frameworks have been synthesized by structure replication from cubic mesoporous KIT-6 silica. The products possess a regular structure with uniform wall thickness (~7 nm) and large internal BET surface area (~87-141 m2/g). The pore walls of these materials consist of nanocrystalline Co3O4 and 12-tungstophosphoric acid (HPW) components with different HPW content, i.e. 6, 15 and 36 wt. %. Total X-ray scattering analysis and UV/vis spectroscopy confirmed the Keggin structure of HPW into the mesoporous frameworks. Preliminary catalytic experiments found that Co3O4-HPW composites exhibited a remarkable activity in the direct decomposition of nitrous oxide into N2 and O2.

[37] G.S. Armatas, G. Bilis & M. Louloudi. Highly Ordered Mesoporous Zirconia-Polyoxometalate Nanocomposite Materials for Catalytic Oxidation of Alkenes. J. Mater. Chem., 22, 2997–3005 (2011).
A series of well-ordered mesoporous ZrO2-based heteropoly acid nanocomposite frameworks has been prepared through a surfactant-assisted sol–gel copolymerization route. The pore walls of these materials consist of nanocrystalline tetragonal ZrO2 and Keggin-type 12-phosphomolybdic acid (PMA) components with different PMA loadings, i.e. 12, 22 and 37 wt%. Small angle X-ray scattering, high-resolution TEM and N2 physisorption measurements indicated mesoporous property in hexagonal p6mm symmetry with large internal BET surface areas and narrow-sized pores. The incorporated PMA clusters preserve intact their Keggin structure into the mesoporous frameworks according to EDX, FT-IR and diffuse-reflectance UV/vis/NIR spectroscopy. The obtained ZrO2–PMA nanocomposites demonstrated great application potential in oxidative catalysis, exhibiting exceptional stability and catalytic activity in oxidation of alkenes using hydrogen peroxide as oxidant.
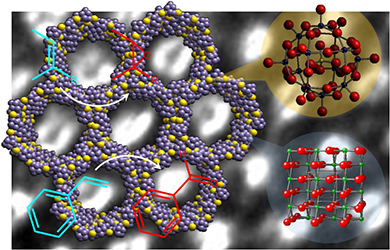
[36] G.S. Armatas* & M.G. Kanatzidis*. Germanium-Based Porous Semiconductors from Molecular Zintl Anions. Structure & Bonding, Thomas F. Fässler (Ed.) in "Zintl Phases and Ions", Springer, Berlin – Heidelberg, 140, 133-154 (2011).
This review highlights how molecular Zintl compounds can be used to create new materials with a variety of novel opto-electronic and gas absorption properties. The generality of the synthetic approach described in this chapter on coupling various group-IV Zintl clusters provides an important tool for the design of new kinds of periodically ordered mesoporous semiconductors with tunable chemical and physical properties. We illustrate the potential of Zintl compounds to produce highly porous non-oxidic semiconductors, and we also cover the recent advances in the development of mesoporous elemental-based, metal-chalcogenide, and binary intermetallic alloy materials. The principles behind this approach and some perspectives for application of the derived materials are discussed.
[35] G.S. Armatas*, A.P. Katsoulidis, D.E. Petrakis, P.J. Pomonis & M.G. Kanatzidis. Nanocasting of Ordered Mesoporous Co3O4-based Polyoxometalate Composite Frameworks. Chem. Mater., 22, 5739–5746 (2010).
We report the synthesis of highly ordered mesoporous frameworks consisting of nanocrystalline Co3O4 and Keggin-type tungstophosphoric acid (HPW12O40, HPW) compounds using the hard-templating method. The resulting materials feature a Co3O4/HPW solid solution structure with different HPW loadings, i.e., 6, 11, 15, and 36 wt %. Characterization by small-angle X-ray scattering (SAXS), high-resolution transmission electron microscopy (TEM), and N2 physisorption measurements reveal that all mesoporous frameworks possess a three-dimensional cubic symmetry with large internal Brumauer−Emmett−Teller (BET) surface area (87−141 m2g−1) and narrow sized pores (ca. 4 nm). The Keggin structure of the incorporated PW12O403− clusters within the composite frameworks was confirmed with X-ray diffused scattering and atomic pair distribution function (PDF) analysis, X-ray photoelectron spectroscopy (XPS), infrared (IR), and diffuse-reflectance UV/vis spectroscopy. Catalytic studies have indicated that these Co3O4/HPW composites can be effective catalysts, exhibiting remarkable catalytic activity on direct decomposition of N2O.
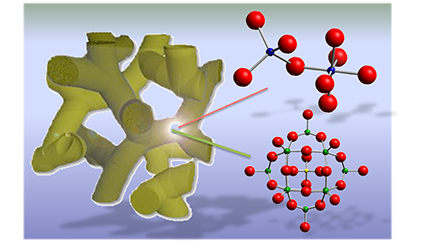
(*Highlighted by ChemInform, 41, 52, 2010)
[34] G.S. Armatas*, A.P. Katsoulidis, D.E. Petrakis & P.J. Pomonis. Synthesis and Acidic Catalytic Properties of Ordered Mesoporous Alumina-Tungstophosphoric acid Composites. J. Mater. Chem., 20, 8631–8638 (2010).
A series of well-ordered mesoporous alumina–tungstophosphoric (HPW) acid composite frameworks has been prepared by a sol–gel copolymerization route in the presence of non-ionic surfactants. The resulting materials feature a high loading of HPW acids (up to ∼53 wt%) in composite framework and possess hexagonal p6mm pore structure with uniform large pores. The mesoscopic order of these structures was evidenced by SAXS analysis, TEM images and N2 physisorption measurements. The composite materials exhibited BET surface areas in the range of 54–71 m2 g−1, total pore volumes in the range of 0.11–0.14 cm3 g−1 and quite narrow pore size distributions with peak maxima in the 7.1–8.3 nm range. The Keggin clusters were incorporated in mesoporous alumina walls by strong chemical bonds according to the FT-IR and UV/vis spectroscopy analysis. This chemical linkage of HPW to the alumina matrix is responsible for the outstanding stability of these materials against water-leaching. The mesoporous surfaces exhibited exceptional acidity that arises from the unique alumina–HPW composite structure. As the loading of HPW increases, the surface acidic character of the composites enhanced, and this is reflected in the higher catalytic activity towards isopropanol conversion.

[33] G.S. Armatas & M.G. Kanatzidis. Size Dependence in Hexagonal Mesoporous Germanium: Pore Wall Thickness versus Energy gap and Photoluminescence. Nano Lett., 10, 3330–3336 (2010).
A series of hexagonal mesoporous germanium semiconductors with tunable wall thickness is reported. These nanostructures possess uniform pores of 3.1−3.2 nm, wall thicknesses from 1.3 to 2.2 nm, and large internal BET surface area in the range of 404−451 m2/g. The porous Ge framework of these materials is assembled from the templated oxidative self-polymerization of (Ge9)4− Zintl clusters. Total X-ray scattering analysis supports a model of interconnected deltahedral (Ge9)-cluster forming the framework and X-ray photoelectron spectroscopy indicates nearly zero-valence Ge atoms. We show the controllable tuning of the pore wall thickness and its impact on the energy band gap which increases systematically with diminishing wall thickness. Furthermore, there is room temperature photoluminescence emission which shifts correspondingly from 672 to 640 nm. The emission signal can be quenched via energy transfer with organic molecules such as pyridine diffusing into the pores.
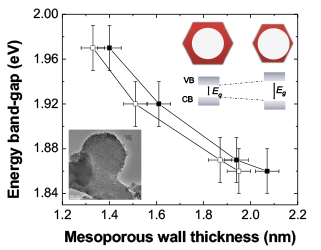
[32] N. Ding, G.S. Armatas & M.G. Kanatzidis. Metal Inorganic Frameworks: Dynamic Flexible Architecture with Extended Pore Order Built from [Se3]2- Linkers and [Re6Se6Br8]2- Clusters. J. Am. Chem. Soc., 132, 6728–6734 (2010).
A mesostructured chalcogenide built from Chevrel-type clusters [(Re6Se6Br2)Br6T]2− linked by ditopic [Se3]2− anions and synthesized in the presence of a cationic surfactant template is reported. This new mesophase, h-C18PyReSeBr, exhibits remarkably well ordered hexagonal symmetry from which a reasonable structural model can be deduced on the basis of powder X-ray diffraction as well as pair distribution function (PDF) analysis. Small-angle X-ray scattering (SAXS) analysis shows that h-C18PyReSeBr possesses an enormous interfacial area of 477 m2/g between the inorganic framework and the guest surfactant cations, which is comparable to that of mesoporous silicas when heavy metals contained in the framework are taken into consideration. The framework of h-C18PyReSeBr exhibits great flexibility and responds dynamically to the extraframework cations via an ion-exchange process.
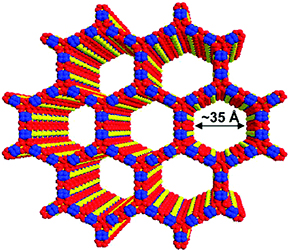
[31] Q. Zhang, G.S. Armatas & M.G. Kanatzidis. Activation of Tellurium with Zintl Ions: 1/∞{[Ge5Te10]4−}, An Inorganic Polymer with Germanium in Three Different Oxidation States. Inorg. Chem., 48, 8665–8667 (2009).
The solvothermal reaction among tellurium, manganese, and the Zintl compound K4Ge9 has led to the formation of a polymeric mixed-valent complex {[Mn(en)3]2(Ge5Te10)}n (1; en = ethylenediamine). The most interesting structural feature of this material is the presence of three different oxidation states of germanium centers and the formation of Ge−Ge bonds in the infinite polymeric chains 1/∞{[Ge5Te10]4−}. X-ray photoelectron spectroscopy characterization confirms the different oxidation states of germanium.
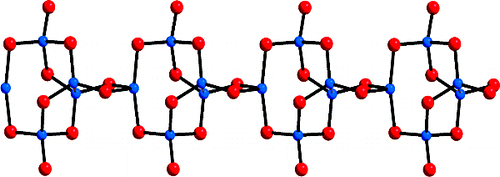
(*Highlighted by ChemInform, 40, 47, 2009)
[30] A.P. Katsoulidis, E.T. Tsaousi, G.S. Armatas, D.E. Petrakis & P. J. Pomonis. Organized Mesoporous Silico-Nickelates (OMSiNi) and Silico-Lanthano-Nickelates (OMSiLaNi): Crystallogenesis vs. Morphogenesis and Microporosity vs. Pore Anisotropy. Micropor. Mesopor. Mater., 122, 175–188 (2009).
In this work organized mesoporous silico-nickelates (OMSiNi) and silico-lanthano-nickelates (OMSiLaNi) have been studied. The synthesis took place in one step using of poly-acrylic acid (Pac), complexed with cetyl-trimethyl-ammonium-bromide (C16TAB) towards a mesostructured flexible backbone on which hydrolysis of tetra-ethyl-ortho-silicate (TEOS) and Ni(NO3)2, or Ni(NO3)2 + La(NO3)3, takes place at increasing pH values. The loading of nickel up to 10%, and nickel plus lanthanum up to 5% each, increases with the pH values (=5.5, 7.5, 9.5) where the materials were isolated and the same effect has the increase of the temperature of the preparation bath from RT to 323 K. The surface area of the OMSiNi and OMSiLaNi solids, containing the lower amounts of metals, was determined in the range ∼1100 and ∼970 m2/g, respectively, and drops as the Ni and/or the Ni + La loading increases. The OMSiNi materials exhibits organized mesoporosity of MCM-41 type in nanometer scale (XRD) and remarkable morphologenesis of the Ni-doped silicate mass in micrometer scale (SEM). The introduction of La in the group OMSiLaNi results in gradual deterioration of organized porosity and destruction of morphogenesis. Both OMSiNi and OMSiLaNi solids contain NiO crystallites of 3–6 nm size (XRD) while the samples with high La content prepared at 323 K show extensive crystallogenesis of perfect NiO crystals of micrometer size (SEM). The pore anisotropy b was determined in the range 7 - b - 56,000 and is lower in the samples prepared at higher temperature and containing higher amounts of metal(s). The % microporosity of the solids is related to the log b by a relationship log b = log b0 − k(% micro) where k is a parameter related inversely to the width of the micropore range of distribution. An explanation of this is proposed based on the assumption that the introduction of micropores, in conjunction with the width of their distribution, result in numerous interruptions of mesopore channels and as a result b decreases exponentially.

[29] G.S. Armatas & M.G. Kanatzidis. Mesoporous germanium-rich chalcogenido frameworks with highly polarizable surfaces and relevance to gas separation. Nat. Mater., 8, 217–222 (2009).
Mesoporous materials with tunable non-oxidic framework compositions can exhibit new kinds of functionality including internal surfaces with high polarizability. As the chemical and physical characteristics of the framework components can induce useful catalytic, absorption and optoelectronic features, the mesoporous structure can promote fast mass diffusion kinetics and size-selective transport of guest molecules1. So far, synthetic efforts have resulted in mesoporous metal chalcogenides on using structure-directing moulds of soft or hard templates. These include ordered mesoporous II–VI semiconductors (such as CdS (refs 2,3), ZnS (ref. 4) and CdTe (ref. 5)). Recently, template-free synthetic routes for high-surface-area chalcogenide aerogels have been reported6,7. Here, we describe a novel kind of porous materials based on germanium-rich chalcogenide networks and ‘soft’ highly polarizable surfaces. We demonstrate that these materials can exhibit excellent selectivity for separating hydrogen from carbon dioxide and methane. These highly polarizable mesoporous structures have important implications for membrane-based gas separation process technologies including hydrogen purification.

(*Highlighted by Science Spot, Extracting the H2, 2009; Alternative energy news, New Hydrogen Purification Method; Science Daily and Northwestern University News, Chemists Offer New Hydrogen Purification Method, 2009)
[28] Y.-M. Jeon, G.S. Armatas, D. Kim, M.G. Kanatzidis & C.A. Mirkin. Tröger’s-base-derived infinite coordination polymer microparticles. Small 5, 46–50 (2009).
A new class of infinite co‐ordination polymer microparticles showing highly selective adsorption of H2 over N2 is developed from the co‐ordination chemistry of acid‐functionalized Tröger's‐base‐derived ligands and Zn(II) metal ions.
[27] G.S. Armatas & M.G. Kanatzidis. Mesoporous compound semiconductors from the reaction of metal ions with deltahedral [Ge9]4- clusters. J. Am. Chem. Soc., 130, 11430–11436 (2008).
We report the surfactant-directed assembly of mesoporous metal/germanium-based semiconducting materials from coupling of anionic (Ge9)4− clusters with various linking metal ions. The resulting materials feature a metal/Ge9 framework perforated by regular arrays of mesoporous channels. The permanent mesoporosity of the materials NU-MGe-2 (M = Sb, In, Sn, Pb, Cd), determined by N2 physisorption measurements, corresponds to high internal BET surface areas from 127 to 277 m2/g and total pore volumes from 0.15 to 0.26 cm3/g. The mesoporous structures exhibit energy gaps in the range of 1.48−1.70 eV as well as strong photoluminescence at room temperature with emission energies varying from 740 to 845 nm. The emission depends on pore wall thickness and framework composition. The photoemission intensity in the mesoporous intermetallic germanium-based frameworks can be selectively suppressed by adsorbing electron-acceptor species such as tetracyanoethylene molecules but remains unchanged when exposed to electron-donor species such as tetrathiafulvalene molecules.
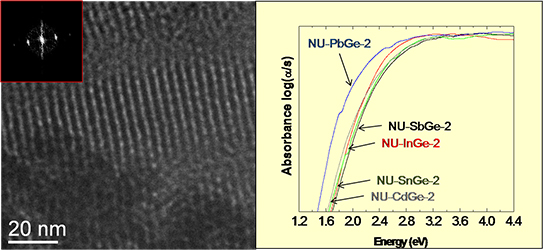
[26] Y.-M. Jeon, G.S. Armatas, J. Heo, M.G. Kanatzidis & C.A. Mirkin. Amorphous Infinite Coordination Polymer Microparticles: A New Class of Selective Hydrogen Storage Materials. Adv. Mater., 20, 2105–2110 (2008).
A new class of micrometer‐sized amorphous infinite coordination particles is selectively prepared from the coordination chemistry of a metallo‐salen building block and Zn2+ ions. The particles show moderately high H2 uptake and almost no N2 adsorption, even though they are amorphous and do not have the well‐defined channels typically used to explain such selectivity in metal–organic framework systems.
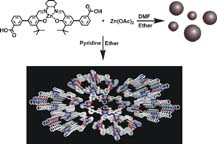
[25] G.S. Armatas & M.G. Kanatzidis. High Surface Area Mesoporous Germanium From The Oxidative Self-Polymerization of The Deltahedral [Ge9]4- Cluster: Electronic Structure Modulation With Donor And Acceptor Molecules. Adv. Mater., 20, 546–550 (2008).
A high‐surface‐area mesoporous germanium is prepared by a new straightforward method that involves oxidative polymerization and no linking agents or external oxidants. The electronic structure of this Ge semiconductor exhibits reversible change towards absorption of various donor and acceptor organic molecules.

[24] S. Bag, P.N. Trikalitis, P.J. Chupas, G.S. Armatas & M.G. Kanatzidis. Porous Semiconducting Gels and Aerogels from Chalcogenide Clusters. Science, 317, 490–493 (2007).
Inorganic porous materials are being developed for use as molecular sieves, ion exchangers, and catalysts, but most are oxides. We show that various sulfide and selenide clusters, when bound to metal ions, yield gels having porous frameworks. These gels are transformed to aerogels after supercritical drying with carbon dioxide. The aerogels have high internal surface area (up to 327 square meters per gram) and broad pore size distribution, depending on the precursors used. The pores of these sulfide and selenide materials preferentially absorb heavy metals. These materials have narrow energy gaps (between 0.2 and 2.0 electron volts) and low densities, and they may be useful in optoelectronics, as photocatalysts, or in the removal of heavy metals from water.
(*Highlighted by ChemInform, 38, 42, 2007; S.L. Brock. Filling a Void, Science, 317, 460, 2007; Aerogel.org, Semiconducting Metal Chalcogenide Aerogels, 2007; Green Car Congress, “Chalcogels”: New Aerogels For Water Decontamination, Hydrogen Purification, 2007; Science Daily, New Aerogels Could Clean Contaminated Water, Purify Hydrogen For Fuel Cells, 2007)
[23] A.E. Giannakas, A.K. Landavos, G.S. Armatas & P.J. Pomonis. Surface properties, textural features and catalytic performance for NO+CO abatement of spinels MAl2O4 (M = Mg, Co and Zn) developed by reverse and bicontinuous microemulsion method. Appl. Surf. Sci., 253, 6969–6979 (2007).
Three typical spinels of general formula MAl2O4 (M = Mg, Co and Zn) have been successfully prepared via a microemulsion method both in the reverse and bicontinuous state. The final solids were characterized by X-ray diffraction followed by Rietveld analysis, N2 adsorption–desorption porosimetry and SEM. Pore connectivity (c) was also calculated with Seaton's method. The analysis of all these properties shown that spinels prepared via reverse microemulsion route have better surface and textural properties than bicontinuous ones. The spinels were tested for NO + CO reaction and reverse spinels shown better catalytic activity than bicontinuous ones while the full sequence of catalytic activity is: ZnAl2O4-r, ZnAl2O4-b, MgAl2O4-r, MgAl2O4-b, CoAl2O4-r, CoAl2O4-b. The Rietveld analysis helped us to give an explanation about the catalytic activity and shown that the configuration of inverse spinel phase is the critical factor for the catalytic behavior of final solids. The reactants NO and CO react in a 2:1 ratio at low temperature but they convert in a 1:1 ratio at high temperatures. From the kinetic analysis the heats of NO adsorption are estimated and are in full agreement with the results of catalytic activity.

[22] A.P. Katsoulidis, D.E. Petrakis, G.S. Armatas & P.J. Pomonis. Microporosity, pore anisotropy and surface properties of organized mesoporous silicates (OMSi) containing cobalt and cerium. J. Mater. Chem., 17, 1518–1528 (2007).
Nanostructured, organized mesoporous silicate materials, containing cerium plus cobalt, were synthesized from a system based on polyacrylic acid (Pac), C16TAB and TEOS. The synthesis took place along a titration path from low pH (≈2) to high pH (≈10). Along this path the C16TAB species are attached to the Pac, and on the formed micelles, hydrolysis of TEOS (as well as of cobalt and cerium species) takes place. The initial atomic ratios of Ce(IV) : Co(II/III) were 1 : 9, 2.5 : 7.5 and 5 : 5. In each of those three cases, three different samples were isolated at pH = 5.5, 7.5 and 9.5 which, after drying and firing at 600 °C, were characterized by X-ray diffraction (XRD), nitrogen adsorption porosimetry, diffuse reflectance spectrospopy (DRS), scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (EDX). Cerium is already quantitatively incorporated into the solid at pH = 5.5. At this low pH value, only ≈20% of the cobalt is incorporated into the solid, but it is fully incorporated at the high pH of 9.5 The silicate species containing a low content of Ce(IV) and Co(II/III) (x : z = 1 : 9) show a self-organized morphology as previously observed in samples free of cerium. This self-organization is destroyed when the content of cerium is increased (x : z = 2.5 : 7.5 and 5 : 5). The samples with a low content of Ce(IV) and Co(II/III) possess an MCM-41 structure, which is gradually degenerated as the amount of the metals increases, and tiny crystallites of CeO2 and Co3O4 become apparent with XRD. The surface area of the solids was in the range 1230–770 m2 g−1, and it drops almost linearly as the content of the metals in the solids increases with the increase of pH. The I-point methodology was successfully applied to detect and distinguish distinct domains of microporosity and mesoporosity in the solids, as well as for the estimation of the percentage of micropore volumes (%Vmic), found in the range 15–30%, using the variation of the C parameter of the BET equation. The pore anisotropy, b, was also determined in the range 10 - b - 2300. A correlation of the form log b = log b0 − (%Vmic) was observed for the materials of the present study, in line with previous findings.
[21] G.S. Armatas & M.G. Kanatzidis. Hexagonal Mesoporous Germanium. Science, 313, 817–820 (2006).
The blending of mesoporosity with the properties of semiconductors promises new types of multifunctional nanomaterials. It would be particularly interesting to combine the shape selectivity of a mesoporous oxide with the electronic and photonic characteristics of a useful semiconductor. We demonstrated the synthesis of a mesoporous germanium semiconductor using liquid-crystals-templated chemistry. The template removal was achieved by a two-step ion-exchange thermal procedure. This semiconductive mesoporous form of germanium possesses hexagonal pore ordering with very high surface area and exhibits strongly size-dependent optical properties as well as photoluminescence.
[20] G.S. Armatas & M.G. Kanatzidis. Mesostructured Germanium with Cubic Pore Symmetry. Nature, 441, 1122–1125 (2006).
Regular mesoporous oxide materials have been widely studied1,2,3,4,5,6,7,8 and have a range of potential applications, such as catalysis, absorption and separation. They are not generally considered for their optical and electronic properties. Elemental semiconductors with nanopores running through them represent a different form of framework material with physical characteristics contrasting with those of the more conventional bulk, thin film and nanocrystalline forms1. Here we describe cubic mesostructured germanium, MSU-Ge-1, with gyroidal channels containing surfactant molecules, separated by amorphous walls that lie on the gyroid (G) minimal surface as in the mesoporous silica MCM-48 (ref. 2). Although Ge is a high-melting, covalent semiconductor that is difficult to prepare from solution polymerization, we succeeded in assembling a continuous Ge network using a suitable precursor for Ge4- atoms. Our results indicate that elemental semiconductors from group 14 of the periodic table can be made to adopt mesostructured forms such as MSU-Ge-1, which features two three-dimensional labyrinthine tunnels obeying space group symmetry and separated by a continuous germanium minimal surface that is otherwise amorphous. A consequence of this new structure for germanium, which has walls only one nanometre thick, is a wider electronic energy bandgap (1.4 eV versus 0.66 eV) than has crystalline or amorphous Ge. Controlled oxidation of MSU-Ge-1 creates a range of germanium suboxides with continuously varying Ge:O ratio and a smoothly increasing energy gap.
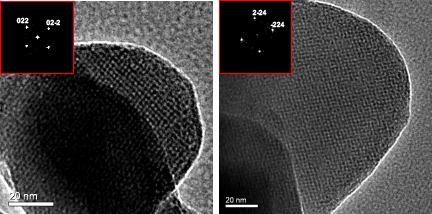
(*Highlighted by ChemInform, 37, 40, 2006; Physics World and Nanotechweb.org, Holey Germanium, 2006; A. Stein. Materials science: Germanium Takes Holey Orders. Nature, 441, 1055, 2006)
[19] G.S. Armatas. Determination of the effects of the pore size distribution and pore connectivity distribution on the pore tortuosity and diffusive transport in model porous networks. Chem. Eng. Sci., 61, 4662–4675 (2006).
An efficient computation method to study flow and transport process of small molecules in porous media using a dual site-bond lattice model, DBSM, is described. The microscopic properties of the porous network take into account the influence of local heterogeneities during the simulations. The numerical experiments demonstrated the combined effect of pore size distribution and connectivity distribution on the mass transport properties and the structural tortuosity. The results indicate that the pore size distribution and percolation phenomena related with pore shielding effects, influence significantly the tortuosity and the effective diffusivity of the porous network. Also, the simulations raise the important role of the connectivity distribution among the various pores in the gas diffusive properties of the poorly connected networks.
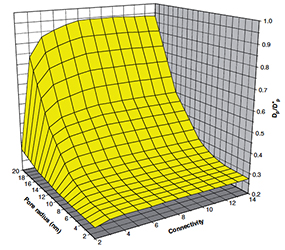
[18] A. Katsoulidis, D.E. Petrakis, G.S. Armatas, P. Trikalitis & P.J. Pomonis. Ordered mesoporous CoOx/MCM-41 materials exhibiting long range self-organized morphology. Micropor. Mesopor. Mater., 92, 71–80 (2006).
A novel class of nanostructured silicious materials of MCM-41 type containing cobalt, CoOx/MCM-41, and exhibiting remarkable long-range self-organized morphology has been developed. The method of synthesis is a one step procedure and includes the use of poly-acrylic acid, complexed with CTAB by a zip-type mechanism, as a backbone of the structure. On the external surface of this mesostructure, hydrolysis of TEOS and Co(NO3)2 takes place at various pH values. The loading of cobalt (9 - Si/Co - 37) in the final solid can be controlled by the pH values at which the material is isolated in the range 5.5 - pH - 9.5. The isolated mesostructures, after drying (80 °C), as well as the resulting mesoporous solids after calcination (600 °C), exhibit remarkable self-organized morphology of the order of μm. In XRD, both the dried mesostructured as well as the calcined solids, exhibit typical patterns of MCM-41 type, which are blurred as the amount of cobalt increases. The surface area of those nanostructured mesoporous solids CoOx/MCM-41 was determined in the range 910–1250 m2/g by nitrogen porosimetry and their pore volume in the range 0.7–0.9 cm3/g. The pore diameter D was found to be around 3.1 nm while the pore anisotropy b = L/D, where L—the length of the pores, was determined in the range 15–380 depending on the sample.

[17] R. Denoyel, J.M. Meneses, G.S. Armatas, J. Rouquerol, K.K. Unger & P.J. Pomonis. Comparative Study of Morphometric Properties Characterizing the Complexity of Silicate Pore Networks Probed by Adsorption of Nitrogen and Methanol. Langmuir, 22, 5350–5357 (2006).
In this work, we compare the surface and morphometric properties of the pore networks in four silicas (code names Fr1428, Fr474, Fr1386, and MM1164) with different random porosities using the adsorption isotherms of two different probe adsorbents, nitrogen and methanol. The parent material Fr1428 was a pure silica 25 μm sample. The Fr474 sample was the same one with bonded electroneutral diol groups on its outer surface. Fr1386 was the parent material with bonded electroneutral diol groups on its outer surface and sulfonic groups on its inner surface, and the MM1164 sample was the original sample with external electroneutral diol groups and internal n-octadecyl groups. The properties examined were the specific surface area Sp and the specific pore volume Vp, the pore connectivity c, the pore anisotropy b, the tortuosity τ, and the lacunarity Λ of the pore network as well as the percentage microporosity. These properties provide a complete characterization of complexity of the porous network. The surface areas of the solids were estimated via the traditional BET plots (SBET) and the I-point method (SI). The two sets of values SBET and SI were practically identical and they decrease as the size of the functional group increases. The values of percentage microporosity were also determined by the same I-point method using the variation of the C parameter of the BET equation. The total pore volume Vp was found to be higher in the case of methanol adsorption, compared to nitrogen, which might be related to increase condensation. The networks of the pores were simulated using a dual site bond model (DSBM) and Monte Carlo (MC) techniques for achieving their proper arrangement into the solids. From the resulting simulating networks, the pore connectivity distributions (PCD) and their mean values cmean were estimated and favorably compared to the values of connectivity cSeaton determined according to the method of Seaton. Both values decrease with the size of the functional groups and are weakly affected by the adsorbent employed. From the simulation pore network, the mean values of tortuosity τmean were also estimated and found to be lower when N2 was used as adsorbate compared to MeOH . The values of lacunarity Λ, estimated according to the method by Allain and Cloitre using the moving box technique in the DSBM/MC simulation matrix of the pore network, indicate that the distribution of the poreless mass into the matrix increases with the size of the functional group. Finally, the internal relationships observed between the pore anisotropy b and the percentage microporosity as well as between the tortuosity τ and the pore connectivity c are discussed.
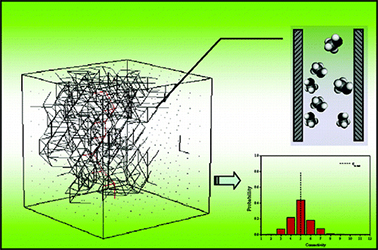
[16] A.E. Giannakas, A.K. Ladavos, G.S. Armatas, D.E. Petrakis & P.J. Pomonis. Effect of composition on the conductivity of CTAB–butanol–octane–nitrate salts (Al(NO3)3 + Zn(NO3)2) microemulsions and on the surface and textural properties of resulting spinels ZnAl2O4. Appl. Surf. Sci., 252, 2159–2170 (2006).
The conductivity σ of a microemulsion series consisting of CTAB + butanol + octane, in which a solution of Al(NO3)3 0.8 M + Zn(NO3)2 0.4 M was gradually added, was studied at room temperature as a function of its composition φ. The addition of nitrate salts solution took place in four different ratios of (butanol + CTAB):octane = 0.2, 0.4, 0.6 and 0.8. Initially, all those four systems are (water in oil, w/o) microemulsions and the gradual addition of the solution of the nitrate salts transforms them to bicontinuous ones. The conductivity increases gradually, but with different rate in each case, and the corresponding critical exponents at the percolation threshold were determined from the curves σ = f(φ). Next at three different compositions of microemulsions, corresponding to ratios (butanol + CTAB):octane = 0.4, 0.6 and 0.8 and ratio of the nitrate salts solution x ≈ 0.25, spinels ZnAl2O4 were isolated/prepared. XRD, SEM and N2 adsorption–desorption measurements were used to determine the structure and texture of those solids. From those measurements the surface area (Sp), the pore volume (Vp), the size of crystallites and the average pore connectivity (c) were found. Those properties showed considerable variation and dependence on the composition of the original microemulsions employed in the preparation, a fact indicating that the structure and texture of the obtained solids can be manipulated at will via the composition of microemulsion used.
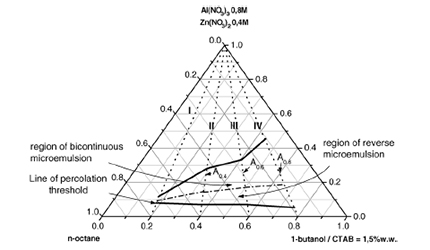
[15] J. Knowles, G.S. Armatas, M. Hudson & P.J. Pomonis. Pore anisotropy and microporosity in nanostructured mesoporous solids. Langmuir, 22, 410–418 (2006).
In this study, we carried out an investigation related to the determination of the anisotropy (b) of pores as well as the extent of microporosity (mic%) in various groups of nanostructured mesoporous materials. The mesoporous materials examined were fifteen samples belonging to the following groups of solids: MCM-48s, SBA-15s, SBA-16s, and mesoporous TiO2 anatases. The porosities of those materials were modified either during preparation or afterward by the addition of Cu(II) species and/or 3(5)-(2-pyridinyl) pyrazole (PyPzH) into the pores. The modification of porosity in each group took place to make possible the internal comparison of the b and mic% values within each group. The estimation of both the b and mic% parameters took place from the corresponding nitrogen adsorption−desorption isotherms. The new proposed method is able to detect a percentage of microporosity as low as a few percent, which is impossible by any of the methods used currently, without the use of any reference sample or standard isotherms. A meaningful inverse relationship is apparent between the b and mic% values, indicating that large values of b correspond to small values of mic%.
[14] G.S. Armatas, D.E. Petrakis & P.J. Pomonis. A Method of Distinction Between Microporosity and Mesoporosity Using BET-Scatchard Plots. Micropor. Mesopor. Mater., 83, 251–261 (2005).
In this work we describe a method for determining microporosity and mesoporosity in porous solids using BET–Scatchard (B–S) plots [P.J. Pomonis, D.E. Petrakis, A.K. Landavos, K.M. Kolonia, G.S. Armatas, S.D. Sklari, P.C. Dragani, A. Zarhala, V.N. Stathopoulos, A.T. Sdoukos, Micropor. Mesopor. Mater. 69 (2004) 97–107]. These plots have the form V[1 − (P/P0)]/(P/P0) = CVm − (C − 1)V[1 − (P/P0)] and provide graphs which have the form of an inclined V. The slope of the lines V[1 − (P/P0)]/(P/P0) vs V[1 − (P/P0)] corresponds to (C − 1) from which the values of C can be determined in the whole range 0 - (P/P0) - 1. The inversion point of those graphs, termed I-point, corresponds exactly to the volume of monolayer Vm which is thus determined independently of C. We employed this technique in 10 silicas with specific surface areas (Sp) varying between 484 and 885 m2 g−1 and, specific pore volume (Vp) between 0.44 and 1.57 cm3 g−1. The percentage of micropore volume (Vmic), determined using as reference standard isotherms and as-plots, was found between 5% and 33% while the corresponding micropore surface area (Smic) was found between 17% and 78%. The same values of Vmic and Smic were determined via the B–S plots, without using any standard isotherms or any non-porous solid as a reference, as follows: First we draw plots of the form C = f(V/Vm) which are unique for each sample. Then we draw the plots dC/d(V/Vm) = f(V/Vm). Those last lines have an S-type form. It was found that the Vmic values estimated from (P/P0) ≈ 0 up to the values of (P/P0) where the lines dC/d(V/Vm) = f(V/Vm) obtain zero slope, correspond to the micropore volume, and tally with the Vmic estimated via the as-plots. This experimental evidence is supported by theoretical suggestions. This new methodology via the B–S plots (I-point method) is easy, unique and self-supported for each particular porous material whose Vmic and Smic values can be thus determined without resolving to the use of any standard isotherm or preparing and testing non-porous samples for comparison.
[13] G.S. Armatas, D.E. Petrakis & P.J. Pomonis. Estimation of Diffusion Parameters in Functionalized Silicas with Modulated Porosity. Part I: Chromatographic Studies. J. Chromatogr. A, 1074, 53–59 (2005).
The diffusion parameters of binary gas mixture He (tracer gas)–N2 (carrier gas) in hybrid organic–inorganic SiO2-X porous solids which have suffered gradual functionalization with functional groups X of increasing length (X = ∅, Si–H, Si–CH2OH, Si–(CH2)3OH, Si–(CH2)11CH3) are reported. The effective diffusivities Deff, the Henry law constants K as well as the tortuosity factors τ for the examined solids were estimated by a typical pulse gas chromatographic method. Analysis of the experimental results was carried out by the well-known method of linearization of moments. The moments s analysis provides a powerful means for extracting diffusion parameters from the experimental response curves The proposed methodology is simple compared to other similar studies and provides rapidly reliable data. The results of this work indicate that the effective diffusivity Deff in porous networks drops markedly as the initial porosity of the parent SiO2 sample is blocked by the functionalization of the pore surfaces with functional groups of increasing size, Si–H, Si–CH2OH, Si–(CH2)3OH and Si–(CH2)11CH3. The low values of the Henry law constants K found indicate that the adsorption of He on the porous surfaces for all the solids is weak. Also, the tortuosity factor τ is proportionally correlated to the pore blocking effects and the percolation phenomena of gases flowing into the porous network.
[12] G.S. Armatas, D.E. Petrakis & P.J. Pomonis. Estimation of Diffusion Parameters in Functionalized Silicas with Modulated Porosity. Part II: Pore Network Modelling. J. Chromatogr. A, 1074, 61–69 (2005).
In this work, the pore structure of those five (5) silicas SiO2–X (see Part I) which have suffered gradual functionalization with functional groups X of increasing length (X = Ø, ≡Si–H, ≡Si–CH2OH, ≡Si–(CH2)3OH, ≡Si–(CH2)11CH3), is modeled as a three-dimensional cubic network of cylindrical pores. Those hybrids organic–inorganic SiO2–X samples are characterized by different extent of pore blocking effects. The pores of samples are represented in a 9 × 9 × 9 lattice by the nodes as well the bonds that are interconnected in a so-called dual site-bond model, DSBM, network. The pore network is developed using a Monte Carlo statistical method where the cylindrical pores (nodes and bonds) are randomly assigned into the lattice, until matching of the theoretical results to the experimental data of N2 adsorption–desorption measurements. Thus, a visual picture of the porous solid is possible. This realistic network is used next in order to study the steady-state gas transport (Knudsen gas-phase and viscous diffusion) properties for the examined materials and how flow processes depend on the morphology of the pore structure. The pore diffusivity Dp and total permeability P of each porous medium is determined based on theoretical calculations and the structural statistical parameters, such as porosity ɛp, tortuosity factor τ and connectivity c of pores is discussed with the corresponding experimental data described in Part I of this work. The results indicate clearly that the diffusion model made it possible to predict pore effective diffusivity in these porous media in very good agreement with the corresponding experimental results for all the examined solids (Part I). The pore diffusivity increases significantly as the value of the pore connectivity increases but the transport properties of the network are influenced strongly at lowest connectivity. Also the predicted tortuosity factor is related inversely to the extent of interconnection of pores in these solids, which indicates that the influence of pore branching to the tortuosity factor of the pore network decreases, as connectivity increases.
[11] G.S. Armatas & P.J. Pomonis. A Monte Carlo Pore Network for the Simulation of Porous Characteristics of Functionalized Silica: Pore Size Distribution, Connectivity Distribution and Mean Tortuosities. Chem. Eng. Sci., 59, 5735–5749 (2004).
The simulation of the random porous network of five samples of functionalized took place using a dual-site-bond model (DSBM) and Monte Carlo techniques for achieving the proper arrangement of the pores into the system. The simulation took place in lattice. As a starting point of simulation the adsorption branch of isotherm was considered from which the pore size distribution was estimated. Then as a benchmark of simulation the desorption branch was considered whose fitting was achieved using the Monte Carlo technique for selecting the proper place of each pore into the lattice. From the obtained model, it was possible to estimate the distribution of pore connectivities of each system. The mean value of those connectivity distributions tallies with the corresponding mean values estimated using the standard methodology of Seaton. In addition, the mean tortuosity of the porous network was estimated and the results were favorably compared with values of tortuosity estimated recently via the so-called corrugated pore-structure-model (CPSM) for the same solids. The degree of functionalization of the parent affects both connectivity and tortuosity in a linear way. Some discrepancies observed between the results obtained via this methodology and the ones obtained using the Seaton or the CPSM model are discussed.
[10] B. Prieto-Simón, G.S. Armatas, P.J. Pomonis, C.G. Nanos & M.I Prodromidis. Metal Dispersed Xerogel Based Composite Films for the Development of Interference Free Oxidase-Based Biosensors. Chem. Mater., 16, 1026–1034 (2004).
We report for the first time the synthesis of metal-modified 3-aminopropyltrimethoxysilanes (APTMS) and their application for the electrochemical monitoring of hydrogen peroxide. Copper-, iron-, zinc-, and cerium-modified APTMS were mixed with tetraethoxysilane (TEOS) and the finally produced xerogels were characterized with scanning electron microscopy, energy dispersive spectrometry, nitrogen porosimetry, and thermal gravimetry. The potential of these materials as catalytic surfaces for the electrochemical monitoring of hydrogen peroxide was investigated by performing cyclic voltammetry and amperometric measurements of metal−APTMS/TEOS modified-platinum electrodes. Results show a significant increase in the sensitivity of the resulting probes compared with the sensitivity of those modified by APTMS/TEOS. However, their applicability and stability were not satisfactory due to the lack of homogeneity, low adhesion onto the electrode surface, and cracking effects during aging. The aforementioned problems were totally eliminated, and the sensitivity of the electrodes was impressively increased, when the metal-modified xerogels were applied over the platinum electrodes as a mixture in a poly(ethylene glycol)−cellulose acetate composite solution (PEG/CA). Apparent diffusion coefficients for different concentrations of PEG were determined with chronocoulometry. Resistance and wettability of the composite films were evaluated with impedance spectroscopy. Metal−APTMS/TEOS−PEG/CA composite films were also successfully tested in the construction of interference-free biosensors for glucose and hydrogen peroxide by immobilizing glucose oxidase and horseradish peroxidase, respectively, into an ethanol-free TEOS sol−gel layer developed over the composite films.
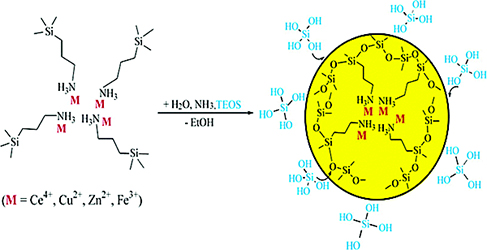
[9] A.A. Leontiou, A.K. Ladavos, G.S. Armatas, P.N. Trikalitis & P.J. Pomonis. Kinetics Investigation of NO+CO Reaction on La–Sr–Mn–O Perovskite-type Mixed Oxides. Appl. Catal. A: Gen., 263, 227–239 (2004).
Substituted perovskite-type mixed oxides of the general formula La1−xSrx(MnIII/MnIV)O3±δ prepared by the nitrate method were examined by X-ray diffractometry (XRD) and the diffraction data were analyzed using the Rietveld refinement method. The amount of desorbed oxygen from the samples when heating in a He atmosphere was estimated with thermogravimetric O2/temperature programmed desorption experiments while the amount of MnIII and MnIV in the samples was determined iodometrically. The substitution of La3+ by Sr2+ leads to a gradual increase of MnIV in the solids and to the transition from an oxygen excess state to an oxygen deficient one. The samples were checked as catalysts for the reaction NO+CO→products in a lab-scale plug–flow reactor between 220 and 560 °C. The reaction rate profiles were simulated using a relationship of the form, where w1, w2 the weighing coefficients for the corresponding R at low Rlow T and high temperatures Rhigh T. From the fitting data the true activation energies E1 and E2 were estimated for Rlow T and Rhigh T as well as the heats of adsorption ΔHads(NO) and ΔHads(CO) for the NO and CO species. Finally, the values of Eapp/low T estimated from typical Arrhenius plots ln R=f(1/T) at the low temperature region are in reasonable agreement with the difference E1−ΔHads(NO), a fact validating the results of simulation/fitting and indicating its usefulness for describing the reaction rates and estimating the kinetic and thermodynamic parameters controlling them. This methodology of simulation provides easily kinetic (activation energies) as well as thermodynamic (heats of adsorption) parameters which otherwise would necessitate differentiated experimental set-ups.
[8] P.J. Pomonis & G.S. Armatas. A Method for the Estimation of Pore Anisotropy in Porous Solids. Langmuir, 20, 6719–6726 (2004).
In this work a method for the estimation of pore anisotropy, b, in porous solids is suggested. The methodology is based on the pore size distribution and the surface area distribution, both calculated from trivial N2 adsorption−desorption isotherms. The materials used for testing the method were six MCM−Alx solids in which the ordered pore structure (for x = 0) was gradually destroyed by the introduction of Al atoms (x = 5, 10, 15, 20, 50) into the solids. Additionally, four silicas having random porosity were examined, in which the surface of the parent material SiO2 (pure silica) was gradually functionalized with organosilicate groups of various lengths (⋮SiH, ⋮SiCH2OH, ⋮Si(CH2)3OH) in order to block a variable amount of pores. As pore anisotropy, the ratio bi = Li/Di is defined where Li and Di are the length and the diameter of each group of pores i filled at a particular partial pressure (Pi/P0). The ratio of the surface area Si over the pore volume Vi, at each particular pressure (Pi/P0), is then expressed as Si3/Vi2 = 16π(Nibi) = 16πλi, where Nibi is the number of pores having anisotropy bi which are filled at each pressure (Pi/P0) and λi is the total anisotropy of all the pores Ni belonging to the group i of pores. Then plot of λi vs (Pi/P0) provides a clear picture of the variation of the total pore anisotropy λi as the partial pressure (Pi/P0) increases. For the functionalized silicas there appears a continuous drop of λi as partial pressure (Pi/P0) increases, a fact indicating that both Ni and bi are continuously diminished. In contrast, for the MCM−Alx materials a sudden kink of λi appears at the partial pressure where the well-defined mesopores are filled up, a fact indicating that at this point Ni and/or bi is large. The kink disappears as the ordered porosity is destroyed by increasing the x doping in MCM−Alx. The pore anisotropy bi of each group i of pores is then estimated using the expression (Si3/Vi2) = 8πNiriSi and plotting log(λi) vs log ri. From those plots, the values of si can be found and therefore the values of bi = 0.5riSi are next defined. In the MCM−Alx materials the maximum pore anisotropy b is very high (bi ∼ 250) for x = 0. Then as mesoporosity is destroyed by increasing x, the maximum b values drop gradually to b ∼ 11 (x = 5), b ∼ 8 (x = 10), and b ∼ 3 (x = 15). For x = 20 and x = 50, the maximum b obtains values equal to unity. The same phenomena, although less profound, are also observed for the functionalized silicas, where the anisotropy b is altered by the process of functionalization and from bi ∼ 0.5 for the nonfunctionalized or bi ∼ 0.9 for the solid functionalized with ⋮SiH groups drops to b = 0.3 and b = 0.2 for the solid functionalized with ⋮Si(CH2)OH and ⋮Si(CH2)3OH, respectively. A correlation factor F is suggested in cases where the pore model departs from the cylindrical geometry.
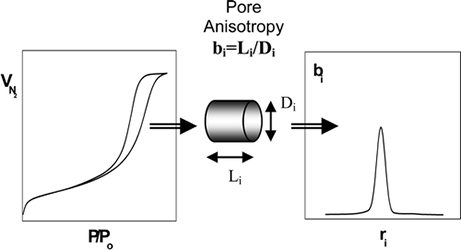
[7] G.S. Armatas & P.J. Pomonis. The Possible Use of Γ–functions for the Determination of Microporosity-Mesoporosity and The Pore Sizes in Materials with Ordered (MCM) and Quasi-Ordered Pore Structure. Micropor. Mesopor. Mater., 67, 167–174 (2004).
The purpose of this work is to examine the possibility to apply Γ-functions for the description of pore size distribution (PSD) as well as for the distinction between the microporosity and mesoporosity in materials with ordered and/or quasi-ordered pore structure. The group of materials used were six (6) MCM-Alx solids (x=0, 5, 10, 15, 20, 50) in which the ordered mesoporosity (x=0) was gradually destroyed by increasing the percentage substitution x of Si by Al. All samples were characterized by N2 adsorption–desorption porosimetry. Then the estimation of PSD took place by using two Γ-functions, one for the micropore region (0-Dp-2 nm) and another for the mesopore region (2-Dp-50 nm). As a benchmark for the choice of Γ-functions their fitting to the N2-adsorption loop was used. Consequently the PSD from those chosen Γ-functions was calculated and the results were compared with the method of Horvath–Kawazoe (HK). The specific surface areas Sp, the specific pore volumes Vp and the maximum Dp of the PSDs calculated by the standard methodologies (HK and BET) are perfectly compared to the ones obtained with the methodology of Γ-functions. In addition the use of Γ-functions methodology provides the possibility to distinguish the percentage of microporosity in such quasi-ordered, amorphous mesoporous, or quasi-mesoporous, materials which is zero for the pure siliceous MCM-Al0 but increases gradually with the addition of Al atoms and reaches ∼32% for the highly substituted solids MCM-Al50.
[6] P.J. Pomonis, D.E. Petrakis, A.K. Ladavos, K.M. Kolonia, G.S. Armatas, S.D. Sklari, P.C. Dragani, A. Zarlaha, V.N. Stathopoulos & A.T. Sdoukos. A Novel method for Estimating the C-values of the BET Equation in the Whole Range 0<P/P0<1 Using a Scatchard – type Treatment of it. Micropor. Mesopor. Mater., 69, 97–107 (2004).
The BET equation, rearranged in a Scatchard-type form, V[1−(P/Po)]/(P/Po) vs. V[1−(P/Po)], was used for the calculation of specific surface areas of a large number (127) of porous materials. The tested solids had surface areas from 20 up to 2200 m2 g−1 and they were of very diverse nature, like clays, mesoporous silicas and aluminas, aluminophosphates with mixed microporosity and mesoporosity, materials with only external porosity based on manganese, zeolites and carbon cloths. The Scatchard-type form of BET equation for all the above solids provide inclined-V type plots, i.e., with an inversion point, termed I-point. The slope of these plots is not constant and corresponds to (C−1) from which the values of C can be estimated in the whole range 0-P/Po-1. Those non-constant C quantities obtain appreciable values (1000–1500) for very low (P/Po). As the I-point is approached at higher (P/Po) the C-values approach the CBET values (C-constant) calculated from the classical BET-plots. Exactly, at the I-point the C-values suffer a transition to infinitive. Then below the I-point, at even higher (P/Po) the (C−1) values become negative. From the corresponding plots it has been possible to calculate C in the whole range of (P/Po) with a notable discontinuity at the I-point. The intercept of the plots V[1−(P/Po)]/(P/Po) vs. V[1−(P/Po)] corresponds to CVm and the Vm value can be estimated if C is known from the slope. Since the slope is not generally constant, this method cannot be used. Nevertheless the I-point corresponds exactly to V[1−(P/Po)]=Vm, i.e. the volume of nitrogen necessary for the formation of a monolayer. The ssa's calculated using this I-point method, are practically identical to those obtained from the classical BET-plots. This methodology provides the possibility to determine the values of the non-constant C-term of the BET equation in the whole range 0-(P/Po)-1 as well as the ssa from the I-point.
[5] G.S. Armatas, C.E. Salmas, M. Louloudi, G.P. Androutsopoulos & P.J. Pomonis. Relationships among Pore Size, Connectivity, Dimensionality of Capillary Condensation and Pore Structure Tortuosity of Functionalized Mesoporous Silica. Langmuir, 19, 3128–3136 (2003).
Seven mesoporous forms of silica were prepared by controlled and gradual functionalization of the original SiO2 surface with silano-(trimethoxy)-propyl-imidazole groups. The degree of surface functionalization was n = 0.00, 0.23, 0.30, 0.40, 0.52, 0.60, and 0.85 and was controlled by previous knowledge of surface acidity, determined by temperature-programmed desorption of NH3. From N2 adsorption/desorption measurements, the specific surface area Sp (m2 g-1), the specific pore volume Vp (cm3 g-1), and the corresponding pore size distributions (PSDs) were determined. The connectivity c of the solids was also calculated according to the method of Seaton, and the dimensionality of capillary condensation Dcc was found using the thermodynamic method of Neimark. The increase of functionalization resulted in a linear drop of Sp and Vp, and the maximum Dmax of the PSD and the full width at half-maximum, fwhm ∼ 2σ, of the distribution drop in a regular way whereas the ratio (Dmax/2σ) remains practically constant. The connectivity c also decreases from c = 12.5 at n = 0 to c = 3 at n = 0.60−0.85, presumably because of blocking of channels connecting various pores. The Dcc values decrease with increasing n values. Next, the co-called corrugated pore structure model, CPSM, was employed for the estimation of tortuosity τ of the porous solids and the simulation of the experimental adsorption/desorption isotherms. From those CPSM simulations, the corresponding specific surface area SCPSM (m2 g-1), the specific pore volume VCPSM (cm3 g-1), and the corresponding pore size distribution PSDCPSM were estimated. The tortuosity τ of the system drops with the degree of functionalization from τ = 4.22 at n = 0 to τ = 3.37 at the initial functionalization (n = 0.23) and subsequently remains practically constant at about τ = 3.40 ± 0.10, for the same reasons which affect the connectivity, that is, blocking of various pore channels. The comparison between the parameters Vp and VCPSM is quite satisfactory. The SCPSM values appear systematically higher by 8−23% compared to the Sp ones. The dimensionality of capillary condensation Dcc is related to the variance 2σ of the PSD. The reasons and the limits of this relationship are discussed.
[4] G.S. Armatas, C.E. Salmas, G.P. Adnroutsopoulos & P.J. Pomonis. The effect of Surface Functionalization of Mesoporous Silicas with Propylimidazol on Porosity, Pore Connectivity and Tortuosity. Stud. Surf. Sci. Cat., 144, 299–306 (2002).
Mesoporous silica was gradually functionalized with silano-(trimethoxy)-propylimidazole (STPI) groups. The degree of surface functionalization n = 0, 0.23, 0.30, 0.40, 0.52, 0.60 and 0.85 was controlled by previous knowledge of surface acidity, determined by TPD/NH3. From N2 adsorption/desorption measurements the specific surface area Sp (m2 g- 1), the specific pore volume VP (cm3 g- 1) and the corresponding pore size distributions PSD were determined while the connectivity c of the solids was calculated according to the method of Seaton. The increase of functionalization resulted in a linear drop of Sp and VP while the maximum Dmax of the PSD and the Full Width at Half Maximum, FWHM ~ 2σ of distribution drop in a regular way whereas the ratio (Dmax/2σ) remains practically constant. The connectivity c also decreases from c = 12.5 at n = 0 to c = 3 at n = 0.60-0.85 presumably because of blocking of channels connecting various pores. Next, the co-called Corrugated Pore Structure Model, CPSM, was employed for the estimation of tortuosity τ of the porous solids and the simulation of the experimental adsorption/desorption isotherms. From those CPSM simulations the corresponding specific surface areas SCPSM (m2 g- 1), specific pore volume VCPSM (cm3 g- 1) and the corresponding pore size distribution PSDCPSM were estimated. The tortuosity τ of the system drops with the degree of functionalization from τ = 4.22 at n = 0 to τ = 3.37 at functionalization n = 0.23 while subsequently remains practically constant at about τ = 3.40 ± 0.10, for the same reasons which affect the connectivity i.e. blocking of various pore channels. The comparison between the parameters VP on one hand and VCPSM, is quite satisfactory while the SCPSM values appear systematically higher by almost 8-23% compared to the Sp ones. Similarities and discrepancies between the results obtained by the two methods are discussed.

[3] G.S. Armatas, K.M. Kolonia & P.J. Pomonis. Morphometry of Porous Solids – Lacunarity, Fractal Dimensions, Connectivity – and Some Topological Similarities with Neurons. Langmuir, 18, 10421–10429 (2002).
The topological morphometry of 16 mesoporous phosphoro−vanado−aluminate solids possessing random porous network and 5 Al-modified MCM 48 materials, possessing ordered porosity, were investigated using the parameters lacunarity (L), fractal dimension (Dv) and connectivity (c) of their porous structure. The phosphoro−vanado−aluminate group of materials employed had the general formula Al100PXVY, with X, Y = 0, 5, 10, 20, the balance being oxygen. The Al−MCM 48 solids contained Al in 0, 5, 10, 15, and 20%. The porosities of the solids were determined using standard N2 adsorption−desorption measurements. The BJH methodology was employed for the determination of the pore size distribution (psd) curves for the Al100PXVY solids while for the Al−MCM 48 materials the psd was estimated using the Howarth−Kawazoe method. From the psd curves, the L of the solids was determined using the formula L = M(2)/[M(1)]2, suggested previously by Allain and Cloitre, where M(1) and M(2) are the first- and second-order momenta of distribution. The Dv of the solids was estimated from plots of the form ln Vp = f[ln(ln(Po/P))] suggested originally by Avnir and Jaroniec, where Vp (cm3 g-1) corresponds to the specific pore volume of the porous materials. Finally the connectivity c of the porous network was determined according to the method of Seaton. The quantities L and c for all materials were found to be interrelated via the relationship: ln L = 0.24 − 1.62 ln c, while the quantities Dv and L are interrelated via the equation Dv = 2.47 − 1.41L. Of interest is the fact that a relationship between D and L, similar to that described above for porous networks, has also been observed previously by Smith and Lange for neurons. The physical meaning underlying this kind of heterosimilarity is discussed from the point of view of natural necessity imposed on the development of such seemingly dissimilar systems that is pores and neurons.
[2] P.J. Pomonis, K.M. Kolonia & G.S. Armatas. Relationship between Pore Connectivity and Mean Pore Size in Modulated Mesoporous Vanado-Phosphoro-Aluminates and Some Similarities with the Branching of Trees. Langmuir, 17, 8397–8404 (2001).
Sixteen mesoporous vanado-phosphoro-aluminate solids, of the general formula Al100PXVY (X, Y = 0, 5, 10, 20), have been prepared and characterized by their N2 adsorption/desorption isotherms. The materials were truly mesoporous, and the addition of P and/or V affects in a very precise and profound way the corresponding specific surface areas Sp (m2 g-1), specific pore volumes (cm3 g-1), the mean hydraulic pore diameter dp (4Vp/Sp) (nm), the maximum of the pore size distribution (psd) dmax (nm), and the full width at half-maximum (fwhm) (nm) of the psd. The pore connectivity c was also determined and the dimensionality Dcc of adsorption was estimated in the pressure range where capillary condensation of N2 in the pores takes place. The dimensionality Dcc was found to be related to the dmax, reduced over the fwhm, via the relation Dcc ≈ 3.7(dmax/fwhm). The two parameters, dimensionality Dcc and connectivity c, are also related to each other via the simple relation log c ∝ −1.83 log Dcc, which in turn results in the relation log c ∝ −1.83 log(dmax/fwhm). A parallelism is drawn between the connectivity of pores in solids and its relation to the pore size, on one hand, and the degree of branching b of trees and its relation with the branch diameter dk, on the other. Using allometric relations already established for such hierarchically developed natural systems, that is, trees, we reach a relation between b and dk of the form log b ∝ −2.34 log(dk,max/2dmean) where dk,max is the average diameter of the higher order (thinner) branches in a typical tree and dmean is the arithmetic mean diameter of all branches of that tree. The reasons for these similarities are discussed.
[1] V.C. Belessi, A.K. Ladavos, G.S. Armatas & P.J. Pomonis. Kinetics of methane oxidation over La-Sr-Ce-Fe-O mixed oxide Solids. Phys. Chem. Chem. Phys., 3, 3856–3862 (2001).
A kinetic study of methane combustion over La0.7Ce0.3FeO3, La0.7Sr0.3FeO3 and La0.7Sr0.1Ce0.2FeO3 mixed oxide solids has been carried out. From kinetic analyses of reaction rate, the combustion can be expressed by the Rideal–Eley mechanism, in which adsorbed oxygen, in dissociative form, is assumed to react with gaseous methane. True activation energies Etrue, in the range 63–75 kJ mol−1, and heats of adsorption of oxygen λO2, in the range 53–211 kJ mol−1, have been determined. The large values of λO2, are related to existence of a SrFeO3±x perovskite crystal phase, which is able to uptake large amounts of oxygen.
Inorganic Chemistry (EMY-221)
Lecture 1. Periodic Table
Lecture 2. Acids-Bases
Lecture 3. Electrochemistry
Lecture 4. Electronic Configuration and Chemical Bonds
Lecture 5. Coordination Chemistry I
Lecture 6. Coordination Chemistry II
Lecture 7. Reactions, Kinetics and Mechanisms of Coordination Complexes
Lecture 8. Solid State Chemistry
Supplementary Notes:
Exercises - Chapter 1-3 , 4-5 and 5-6
The Hard-Soft Acid-Base Principle , Hard and Soft Acids and Bases , Kinetic Stability of Coordination Compounds
Periodic Table of Elements (pdf) , Περιοδικός Πίνακας Χημικών Στοιχείων
Ονοματολογία Ανόργανων Ενώσεων ![]()
Ύλη προόδου
Αποτελέσματα Προοόδου 2025 ![]()
Solid State Chemistry (EMY-425)
Lecture 1. Crystal Structures and Symmetry
Lecture 2. Crystal Defects and Solid Solutions
Lecture 3. Bonding in Solids
Lecture 4. X-ray Diffraction
Lecture 5. Electron Microscopy
Lecture 6. X-ray Photoelectron Spectroscopy
Lecture 7. Synthesis and Fabrication Methods
Syllabus
Supplementary Notes:
Homework - Crystal Structures , Homework - X-ray Diffraction & Crystal Defects
Introduction to Solid State Chemistry (MIT OpenCourseWare)
Crystallography - CSIC , Crystallographic CourseWare (Bucknell)
Symmetry Resources (Otterbein University) , Symmetry and Point Group
,
Miller Indices
Solid State Structures Based on Close Packing (University of Sydney)
Solid State Structures Based on Close Packing
Materials Chemistry Laboratory (ETY-225)
Lecture 1. Superconductor YBa₂Cu₃O₇
Lecture 2. Zeolite NaX
Lecture 3. Nanoparticles CdS
Lecture 4. [Co(NH₃)₄CO₃]NO₃ and [Co(NH₃)₅Cl]Cl₂ Compounds
Εργαστηριακές Ασκήσεις:
Άσκηση 1 , Άσκηση 2 , Άσκηση 3 , Άσκηση 4
Άσκηση 5 , Άσκηση 6 , Άσκηση 7 , Άσκηση 8
Κανονισμός εργαστηρίου
 Laboratory Safety Rules
Laboratory Safety Rules
Transmission Electron Microscopy (METY-446)
Lecture 1. Introduction to TEM
Lecture 2. Electron scattering and diffraction
Lecture 3. Electron guns and electron lenses
Lecture 4. The TEM instrument
Lecture 5. Pums and holders & Sample preparetion
Lecture 6. Crystallography & Electron diffraction * (Lectures by Prof. Pantelis Trikalitis)
Syllabus
Useful links:
Introductory courses on TEM (MATTER - University of Liverpool)
Tutorial courses in TEM (John Rodenburg)
Electron Microscopy Image Simulation (EMS on-Line)
Advanced Solid-State Materials and Nanostructures (METY-501)
Προγραμμα Επικαιροποιησης Γνωσεων Αποφοιτων (ΠΕΓΑ) ΑΕΙ

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|

|
|
Our research Laboratory is well-equipped with a variety of characterization equipment and instrumentation for synthesis of materials, including nanomaterials, catalysts and adsorbents. The group also has use Materials Science and Engineering department research facilities (TGA/DSC, UV-vis, FT-IR) and School of Sciences and Engineering (UOC) analytic services (TEM, FE-SEM, XRD, high-energy X-ray total scattering & pair distribution function analysis (HEXTS/PDF), XPS, Solid-state NMR, TR-PL, FT-Raman).
